What is leucine zipper transcription factor?
The basic leucine zipper (bZIP) transcription factors are sequence-specific DNA-binding proteins that regulate transcription. They are characterized by a 60-80 amino acid bZIP domain: a basic DNA binding domain followed by a leucine zipper dimerization domain.
Why is it called a leucine zipper?
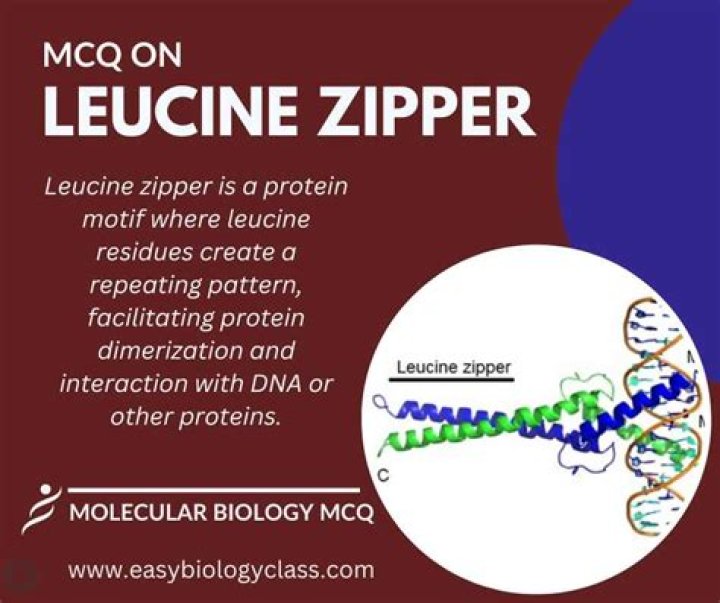
This class of DNA binding proteins gets its name from the regular pattern of leucine residues within the two alpha helices (pictured below on the right). Being hydrophobic, the leucines cause two adjacent alpha helices to be “zippered” together by hydrophobic interactions.
Do leucine zippers bind to DNA?
Basic leucine zipper (bZIP) and basic helix-loop-helix (bHLH) The bZIP and bHLH proteins bind DNA as obligate dimers. The basic region of each monomer forms a helix that contacts DNA in a manner somewhat similar to that of the HTH recognition helix.
How are leucine zippers held together?
A leucine zipper is formed by two α helices, one from each monomer. The helices are held together by hydrophobic interactions between leucine residues, which are located on one side of each helix.
How does a leucine zipper work?
Leucine zipper is created by the dimerization of two specific alpha helix monomers bound to DNA. These amino acids are spaced out in each region’s polypeptide sequence in such a way that when the sequence is coiled in a 3D alpha-helix, the leucine residues line up on the same side of the helix.
Why is leucine zipper stable?
The leucine zipper is a dimeric coiled-coil protein structure composed of two amphipathic alpha-helices with the hydrophobic surfaces interacting to create the dimer interface. Several workers have reported that amino acids in the e and g positions of the coiled coil can modulate dimerization stability and specificity.
Which one of the following contains a leucine zipper motif?
Explanation: The B-ZIP (basic-region leucine zipper) class of eukaryotic transcription factors contain a leucine zipper DNA-binding motif. It does not contain homeodomain or zinc fingers or an H-T-H motif. 11. The leucine zipper is a left-handed parallel dimeric coiled-coil.
What are basic leucine zipper proteins?
Basic leucine zipper (bZIP) proteins are a class of transcription factors characterized by a basic leucine zipper motif, which allows for both dimerization and sequence-specific DNA-binding interactions ( Ellenberger, 1994 ). Transcription factors containing bZIP domains are found across eukaryotes, from budding yeast to humans.
How do leucine zipper and helix-loop-helix motifs act on DNA?
Both the leucine zipper and the helix-loop-helix motif therefore act by causing dimerization, allowing DNA binding by the adjacent basic motif. Interestingly, the Myc oncoproteins contain both a helix-loop-helix motif and a leucine zipper region adjacent to the basic DNA binding region (Landshultz et al., 1988; Murre et al., 1989a ).
Which eukaryotic transcription factors contain bZIP domains?
Transcription factors containing bZIP domains are found across eukaryotes, from budding yeast to humans. Both KSHV and EBV encode bZIP proteins that function as transcriptional modulators during lytic infection ( Table I ).
How does the ZIP domain interact with the DNA sequence?
These basic residues interact in the major groove of the DNA, forming sequence-specific interactions. The leucine zipper is formed by amphipathic interaction between two ZIP domains. The ZIP domain is found in the alpha-helix of each monomer, and contains leucines, or leucine-like amino acids.



