What is limitation reagent?
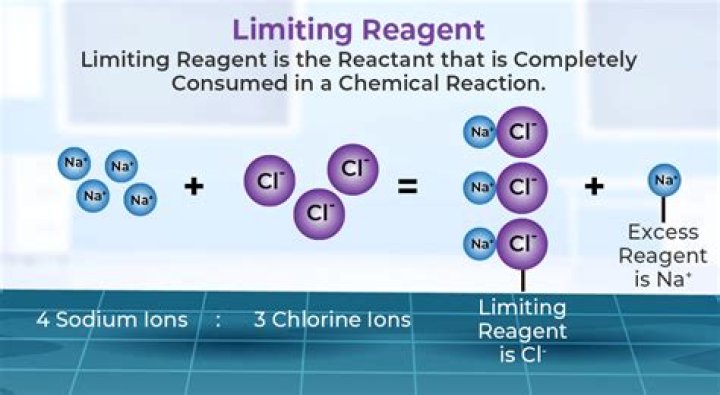
The limiting reagent (or limiting reactant or limiting agent) in a chemical reaction is a reactant that is totally consumed when the chemical reaction is completed. The amount of product formed is limited by this reagent, since the reaction cannot continue without it.
How do you find the limiting reagent in Class 11?
The first step is to balance the chemical equation for the given chemical reaction. Then, convert the given information into moles. Use stoichiometry for each individual reactant for finding the mass of product produced. The reactant which produces a lesser amount of product would be the limiting reagent.
What is limiting reagent and excess reagent?
In a chemical reaction, reactants that are not used up when the reaction is finished are called excess reagents. The reagent that is completely used up or reacted is called the limiting reagent, because its quantity limit the amount of products formed.
What is limiting reagent 11th?
Limiting Reagent. Limiting Reagent. In a chemical reaction, reactant which is present in the lesser amount gets consumed after sometime and after that no further reaction takes place whatever be the amount of the other reactant present.
What is excess and limiting reagent?
The limiting reagent in a chemical reaction is the reactant that will be consumed completely. Therefor it limits the reaction from continuing. Excess Reagent. The excess reagent is the reactant that could keep reacting if the other had not been consumed.
What is limiting reagent in Class 11?
The reactant which reacts completely in the reaction is called limiting reactant or limiting reagent. The reactant which is not consumed completely in the reaction is called excess reactant .
What is limiting reagent class11?
In a chemical reaction, reactant which is present in the lesser amount gets consumed after sometime and after that no further reaction takes place whatever be the amount of the other reactant present.
What is limiting reagent for Class 12?
What is Limiting Reagents? The reactant that is entirely used up in a reaction is called as limiting reagent. In the reaction given above, 3 moles of Hydrogen gas are required to react with 1 mole of nitrogen gas to form 2 moles of ammonia.
How does one determine which reagent is a limiting reagent?
Answer. In order to determine which reactant is the limiting reagent, take each reactant separately and assume that it is the limiting reagent. The reactant that produces the least amount of product must be the limiting reagent.
How do you calculate limiting reactants?
To determine the limiting reactant, calculate the amount of product formed by each reactant. The reactant the produces the least amount of product is the limiting reactant. To determine the number of grams of Na3PO4 formed: The sodium hydroxide formed less product than the phosphoric acid.
How do you find the limiting reagent?
How to Find the Limiting Reagent: Approach 1. Find the limiting reagent by looking at the number of moles of each reactant. Determine the balanced chemical equation for the chemical reaction. Convert all given information into moles (most likely, through the use of molar mass as a conversion factor).
How to calculate limiting reactant.?
Balance the chemical equation.



