What is meant by isotopic fractionation?
isotopic fractionation, enrichment of one isotope relative to another in a chemical or physical process. Two isotopes of an element are different in weight but not in gross chemical properties, which are determined by the number of electrons.
What is isotopic fractionation used for?
Fractionation ratios and isotopic ratios are useful in determining palaeotemperatures, geologic processes, and the modes of formation of rocks and minerals. See D/H RATIO; OXYGEN-ISOTOPE RATIO; OXYGEN-ISOTOPE ANALYSIS; STABLE-ISOTOPE STUDIES; and ISOTOPE GEOCHEMISTRY.
What is the principle of isotope fractionation technique?
Isotopic fractionations are related to bond strength. Thus, stronger bonds generally favor heavier isotopes. A difference in the bond strengths of complexes in a ligand exchange reaction thus results in isotopic fractionation.
Why does isotope fractionation occur?
In every chemical or physical reaction, e.g. oxidation or evaporation, the light isotopes react at a slightly higher rate than heavy ones, because the bonds in a molecule or crystal between lighter atoms vibrate with a higher frequency and therefore they split easier than those between heavier atoms.
How do you calculate isotopic composition?
Then add. Example: Calculate the relative atomic mass (Mr) of chlorine from the 35Cl (75.78%) and 37Cl (24.22%). Method 2 (easier in practice): Multiply each percentage by the mass number….How can we use the isotopic composition to calculate the relative atomic mass?
| Mr(Cl) = | (75.78 × 35 + 24.22 × 37) | = 35.48 |
|---|---|---|
| 100 |
What are the uses of isotopes of hydrogen?
Deuterium and its compounds are used as a non-radioactive label in chemical experiments and in solvents for 1H-NMR spectroscopy. Heavy water is used as a neutron moderator and coolant for nuclear reactors. Deuterium is also a potential fuel for commercial nuclear fusion.
What are the processes that control the isotopic fractionation of oxygen and hydrogen in water?
What climate factors influence the ratio of oxygen isotopes in ocean water? Evaporation and condensation are the two processes that most influence the ratio of heavy oxygen to light oxygen in the oceans. Water molecules are made up of two hydrogen atoms and one oxygen atom.
What is isotopic composition?
In principle, the simplest way to indicate the isotopic composition of an element is to give the abundance of each isotope in atom %. For instance, about 1 % of all carbon atoms on Earth is 13C or heavy carbon; about 0.36 % of all nitrogen atoms is heavy nitrogen (15N).
What is tritium used for?
What arc the uses of tritium? Tritium has been produced in large quantities by the nuclear military program. It is also used to make luminous dials and as a source of light for sarety signs. Tritium is used as a tracer for biochemical research, animal metabolism studies and ground water transport measurements.
What is the importance of heavier isotopes of hydrogen?
an atom of tritium has one proton and two neutrons is its nucleus. tritium atoms are unstable and are radioactive whereas the former two are non-radioactive. It is found in heavy water. heavy water is used to control the nuclear reaction in nuclear reactor and helps to slow down the speed of neutrons.
What is a practical use for o18?
O-18 is used as a tracer in many hydrologic studies. It is most often used as a component in a mixing-model and hydrograph separation, as O-18 acts conservatively and is applied naturally and uniformly over broad areas. O-18 can be used when different sources (old water/new water) have different isotopic values.
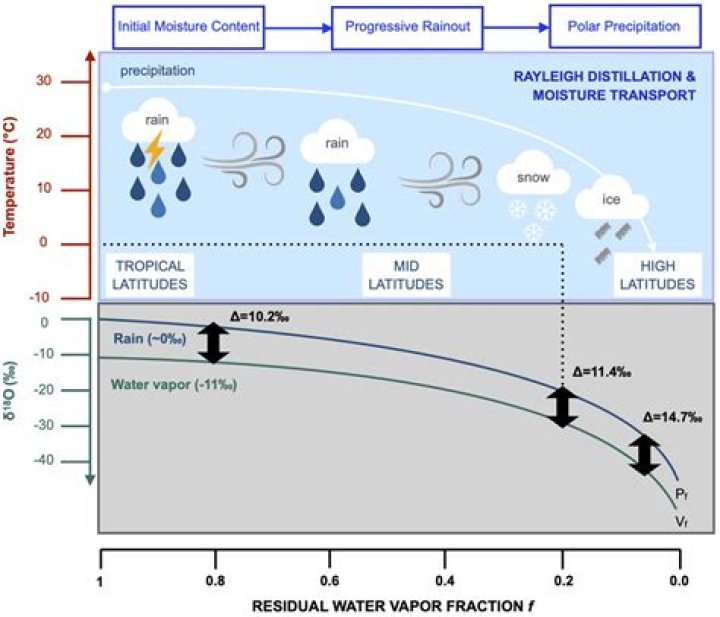
Isotope fractionation occurs during a phase transition, when the ratio of light to heavy isotopes in the involved molecules changes. When water vapor condenses (an equilibrium fractionation), the heavier water isotopes (18O and 2H) become enriched in the liquid phase while the lighter isotopes (16O and 1H) tend toward the vapor phase.
What is the equation for isotopes?
Enter the values into the following formula: a = b (x) + c (1 – x). In the equation, “a” is the average atomic mass, “b” is the atomic mass of one isotope, “c” is the atomic mass of the other isotope, and “x” is the abundance of the first isotope.
How do you calculate the atomic mass of isotopes?
To calculate the atomic mass of a single atom of an element, add up the mass of protons and neutrons. Example: Find the atomic mass of an isotope of carbon that has 7 neutrons. You can see from the periodic table that carbon has an atomic number of 6, which is its number of protons.
What is the fractional abundance of an isotope?
Fractional Abundance or % Natural Abundance means the same. It refers to the abundance of isotopes of a chemical element as naturally found on a planet. The fractional abundance normally represented as a percentage, of each isotope of a given element in nature.



