What is polymeric backbone in Phosphazenes?
The phosphazene polymers constitute a family of greatly diverse performance materials. These polymers possess a ‘backbone’ of alternating nitrogen and phosphorous atoms. To a basic ‘backbone’ polymer, a variety of substituents can be added that control the physical properties of the final product.
What are polymeric backbone in silicones and Phosphazenes?
Silicones are inorganic polymers, that is, there are no carbon atoms in the backbone chain. The backbone is a chain of alternating silicon and oxygen atoms. Each silicone has two groups attached to it, and these can be any organic groups. This polymer is called polydimethylsiloxane.
What is the structural unit of all Phosphazenes?
Phosphazenes refer to classes of organophosphorus compounds featuring phosphorus(V) with a double bond between P and N. One class of phosphazenes have the formula RN=P(NR2)3. These phosphazenes are also known as iminophosphoranes and phosphine imides.
What are Phosphazenes how they are prepared?
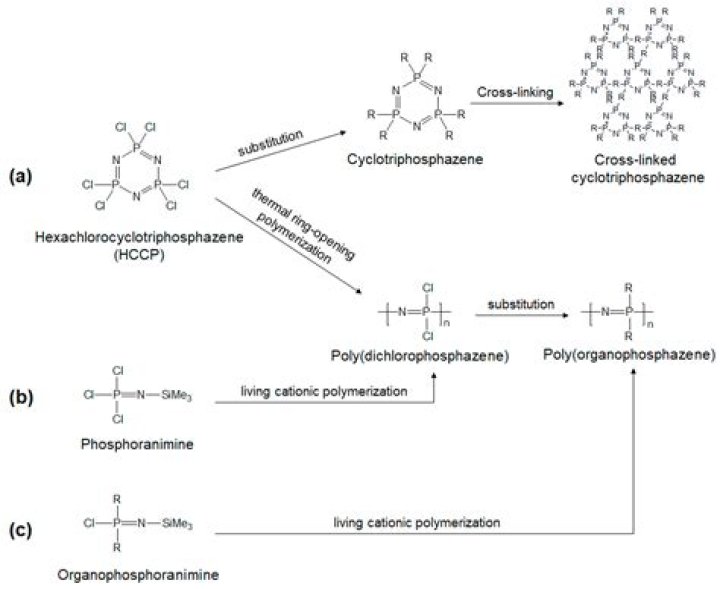
The poly(dichlorophosphazene) (PZ) was obtained by melt ring-opening polymerization of hexachlorocyclotriphosphazene (HCCP) under vacuum at 250°C for 3 h. After this time, the polymer was dissolved at room temperature in anhydrous THF, and it was separated by precipitation into n-heptane.
Why Phosphonitrile are known as Phosphazenes?
*Phosphazenes are a class of chemical compounds in which a phosphorus atom is covalently linked to a nitrogen atom by a double bond and to three other atoms or radicals by single bonds. *Phosphazenes are also knownas iminophosphoranes and phosphonitriclic imides.
What is basic unit of Polyphosphazene?
1.16. Polyphosphazenes (PPHOSs) are polymers with an inorganic backbone consisting of alternating phosphorus and nitrogen atoms, separated by alternating single and double bonds, and organic substituents (R) covalently bonded to the phosphorus atoms as side groups (see Scheme 1.44).
What do you mean by Phosphazenes?
Phosphazenes are a class of chemical compounds in which a phosphorus atom is covalently linked to a nitrogen atom by a double bond and to three other atoms or radicals by single bonds. Phosphazenes are also known as iminophosphoranes and phosphine imides. The corresponding polymers are polyphosphazenes.
What are Phosphazenes discuss the nature of bonding in Triphosphazenes?
The bonds between the phosphorus and nitrogen in phosphazenes are best described as having a significant ionic component with each of the N atoms having a negative charge and each of the phosphorus atoms having a positive charge.
What are Phosphazenes inorganic chemistry?
Phosphazenes, which are cyclic or linear chain inorganic compounds formed by the bonding and repetition of phosphorus and nitrogen atoms with (P=N)n bonds, comprise an important class of inorganic compounds (Figure 1). There are many phosphazene compounds ranging from oligomers to polymers.
What are polyphosphazenes write their important uses?
Polyphosphazenes are at the interface between inorganic and organic polymers as their backbone consists of nitrogen–phosphorus bonds. The main applications of polyphosphazenes are in the field of drug delivery and in skeletal tissue regeneration.
What are organo Phosphazenes?
1.1 Background. Polyphosphazenes are macromolecules with a phosphorus–nitrogen backbone, substituted by two (commonly organic) side groups on the phosphorus atoms to give poly(organo)phosphazenes (Fig. 1).
Which Phosphazenes are more stable?
Bookmarked Docs
- Phosphazenes are believed to be more stable that benzene because its pi delocalised electrons are from p and d orbitals while benzene has p orbitals only.
- •Phosphazenes are therefore much more difficult to reduce than benzene.
What is a phosphazene polymer?
The phosphazene polymers constitute a family of greatly diverse performance materials. These polymers possess a ‘backbone’ of alternating nitrogen and phosphorous atoms. To a basic ‘backbone’ polymer, a variety of substituents can be added that control the physical properties of the final product.
Why are polyphosphazene derivatives available?
Because of the endless number of permutations of substituents that can be attached to the basic ‘backbone’ polymer, polyphosphazenes can be customized to suit any client’s performance-polymer needs. ,technically, Incorporated has manufactured polyphosphazene derivatives since 1994.
What is phosphazene ring technology?
Phosphazene rings and polymers provide an almost unique platform for the development of optical materials. The high electron density in the skeleton and the broad window of transparency from the near ultraviolet to the near infrared provides an excellent starting point for the design and synthesis of a variety of optical and photonic materials.
What is the difference between gray spheres and polyphosphazenes?
Gray spheres represent any organic or inorganic group. Polyphosphazenes include a wide range of hybrid inorganic – organic polymers with a number of different skeletal architectures with the backbone P – N -P-N-P-N-. In nearly all of these materials two organic side groups are attached to each phosphorus center.



