What is sodium and chlorine together called?
Sodium chloride, also known as table salt, is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium and chloride ions. It is commonly used as a condiment and food preservative. Salt can be created by adding two very reactive elements together: sodium (Na(s) metal and chlorine (Cl2(g) gas.
What would you see when sodium reacts with chlorine?
Sodium and chlorine react vigorously when heated, giving an orange flame and clouds of white sodium chloride.
Can you mix bleach and salt together?
When dissolved in water, bleach provides OH-. There will be a neutralization reaction if the salt you use is acidic. There could be a reaction if the salt you use is basic. The salt will be dissolved in a bleach solution.
What do sodium and chlorine make when combined?
Sodium and chlorine, two highly reactive elements, combine to form the stable compound sodium chloride (ordinary table salt).
What happens to sodium and chlorine when they form sodium chloride?
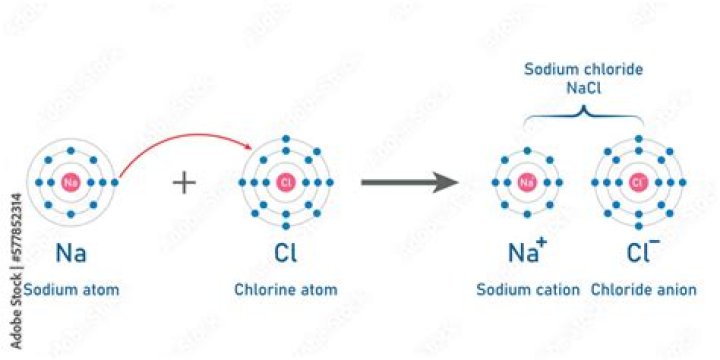
When sodium and chlorine atoms come together to form sodium chloride (NaCl), they transfer an electron. The sodium (Na) atom transfers one electron to the chlorine (Cl) atom, so that they both have full outer shells. When this happens, the atom is called a positive ion.
Why is combined sodium and chlorine safe?
The chlorine has one extra electron and its outer electron shell is complete, so like sodium it too cannot bond with other atoms.
What happens when you mix sodium chloride and water?
When salt is mixed with water, the salt dissolves because the covalent bonds of water are stronger than the ionic bonds in the salt molecules. Water molecules pull the sodium and chloride ions apart, breaking the ionic bond that held them together.
What do you get when you mix salt and bleach?
Ordinary table salt (sodium chloride, NaCl) is half chlorine, and a simple electrochemical reaction with salt water produces chlorine gas easily. That same reaction produces sodium hydroxide (NaOH), and by mixing chlorine gas with sodium hydroxide you create sodium hypochlorite (NaOCl).
Can you mix chlorine and bleach?
The combination produces chlorine gas, as in the chemical warfare agent. Chlorine gas causes coughing and will irritate mucous membranes. It causes chemical burns and can be deadly if concentrations are high enough or exposure is prolonged. Vinegar is not the only acid that produces chlorine gas when mixed with bleach.
Can you separate sodium and chlorine in salt?
Sodium metal and chlorine gas can be obtained with the electrolysis of molten sodium chloride. Electrolysis of aqueous sodium chloride yields hydrogen and chlorine, with aqueous sodium hydroxide remaining in solution.
How is sodium chloride different from sodium and chlorine?
The sodium chloride is an ionic compound formed by the sodium ion and chlorine anion. The sodium atom is a silvery white which vigorously burns in air and is explosive on coming in contact with water whereas the chlorine is a yellow-green gas which corrodes the metal and is poisonous.
Why do sodium and chlorine combine so easily?
Since sodium has exactly one extra of what chlorine so disparately wants, a bond is formed between them with ease. The difference between the strength of the hold that sodium and chlorine have on the bonding electron is so great that the electron pretty much leaves the sodium atom completely and orbits around the chlorine atom alone.
How does sodium react with chlorine?
Explanation of what happens when sodium reacts with chlorine: A sodium atom has one electron in the outer shell. A chlorine atom seven electrons in the outer shell. A sodium atom loses an electron to a chlorine atom. The sodium atom becomes a positive sodium ion. The chlorine atom becomes a negative chloride ion.
What compound does sodium and chlorine make?
Sodium combines with water in an explosive reaction. Chlorine (Cl) is a halogen; it is a highly reactive element that readily gains an electron to fill its outermost shell. Sodium and chlorine, two highly reactive elements, combine to form the stable compound sodium chloride (ordinary table salt).
What elements combine with chlorine?
Nitrogen can combine with three chlorine atoms, forming Nitrogen trichloride, or NCl3. Nitrogen shares its electrons with the chlorine atoms, so all of the atoms have their shells filled. Take a look at the dots around the atoms.



