What is the atomic structure of oxygen?
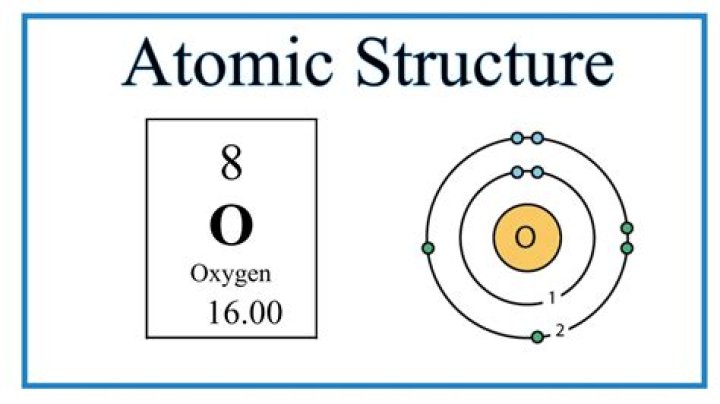
The nucleus of oxygen consists of 8 protons (purple) and 8 neutrons (green). It features eight electrons (yellow) placed on the electron shells (rings).
What is atomic structure answer?
Atomic structure refers to the structure of an atom comprising a nucleus (centre) in which the protons (positively charged) and neutrons (neutral) are present. The negatively charged particles called electrons revolve around the centre of the nucleus.
What is oxygen’s atomic number?
8
Oxygen/Atomic number
What is an atomic structure?
the structure of an atom, theoretically consisting of a positively charged nucleus surrounded and neutralized by negatively charged electrons revolving in orbits at varying distances from the nucleus, the constitution of the nucleus and the arrangement of the electrons differing with various chemical elements.
What is atomic structure?
Atoms consist of three basic particles: protons, electrons, and neutrons. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). The outermost regions of the atom are called electron shells and contain the electrons (negatively charged).
How do we know atomic structure?
The atomic number tells you the number of protons, and if the atom is neutral, the number of electrons, and that electrons are located in the electron cloud surrounding the nucleus. If you know the atomic number of an element, you can determine the number of protons and electrons.
Who discovered oxygen in 1774?
Joseph Priestley
When Joseph Priestley discovered oxygen in 1774, he answered age-old questions of why and how things burn.
What is atomic structure in simple words?
The definition of atomic structure is the positively charged nucleus and the negatively charged electrons circling around it, within an atom. An example of atomic structure is what gives energy to atoms and then to molecules. noun.
What is the color of oxygen?
colorless
The gas is colorless, odorless, and tasteless. The liquid and solid forms are a pale blue color and are strongly paramagnetic.
How do you find the atomic structure?
The symbol for an atom can be written to show its mass number at the top, and its atomic number at the bottom. To calculate the numbers of subatomic particles in an atom, use its atomic number and mass number: number of protons = atomic number. number of electrons = atomic number.



