What is the density of ammonium hydroxide?
880 kg/m³
Ammonia solution/Density
How do you make a 28% ammonium hydroxide solution?
28 wt% (as NH3) can be obtained by either adding water to a 30 wt% (as NH3) sol., or by mixing both 25wt% and 30wt% ammonia solutions. We can retrieve densities of ammonia (aq. sol.)
What is the formula weight of ammonium hydroxide?
35.04 g/mol
Ammonia solution/Molar mass
What molarity is 28 ammonium hydroxide?
Therefore, we can say that 1 liter of Ammonium hydroxide contains 14.534 moles or in other words molarity of 56.6% (w/w) Ammonium hydroxide is equal to 14.534 M….Known values.
| Known values | |
|---|---|
| Density of 56.6% Ammonium hydroxide solution | 0.9 g/ml |
| Molecular weight of NH4OH | 35.05 g/mole |
Is ammonium hydroxide a weak base?
In this Question, Ammonium hydroxide (NH4OH) is called a weak alkali because it is soluble in water & the pH of ammonium hydroxide is in between 7−10 & NH4OH is not completely dissociated into ions. Ammonium hydroxide is also a weak base that does not ionize completely in the given aqueous solution.
How do you make ammonium hydroxide solution?
The Ammonium hydroxide is prepared by saturating water with gaseous ammonia.
What is ammonia density?
0.73 kg/m³
Ammonia/Density
What is the relative formula mass of ammonium hydroxide?
35.046
Ammonium hydroxide
| PubChem CID | 14923 |
|---|---|
| Molecular Formula | H5NO or NH4OH |
| Synonyms | ammonium hydroxide 1336-21-6 Ammonia aqueous Ammonium hydroxide ((NH4)(OH)) Ammonium hydroxide solution More… |
| Molecular Weight | 35.046 |
| Component Compounds | CID 962 (Water) CID 222 (Ammonia) |
Why is ammonium hydroxide not a strong base?
Ammonium hydroxide (NH4OH) is a weak base because the production of the number of OH– ions are very low as compared to the number of NH3 we dissolved in water, this is because ammonia is itself a weak base, and it’s only a tiny fraction of it reacts with water to form(NH4+ and OH–), hence, an only small fraction of OH– …
What is the density of ammonium hydroxide in water?
Ammonia solutions, relative density between 0.880 and 0.957 at 15 C in water, with >10% but not >35% ammonia [UN2672] [Corrosive] Ammonium hydroxide appears as a colorless aqueous solution.
What does ammonium hydroxide look like?
Ammonium hydroxide appears as a colorless aqueous solution. Concentration of ammonia ranges up to approximately 30%. Ammonia vapors (which arise from the solution) irritate the eyes.
Is ammonium hydroxide harmful to eyes?
Ammonia vapors (which arise from the solution) irritate the eyes. Ammonium hydroxide is a solution of ammonia in water. It has a role as a food acidity regulator. azanium;hydroxide Computed by Lexichem TK 2.7.0 (PubChem release 2021.05.07)
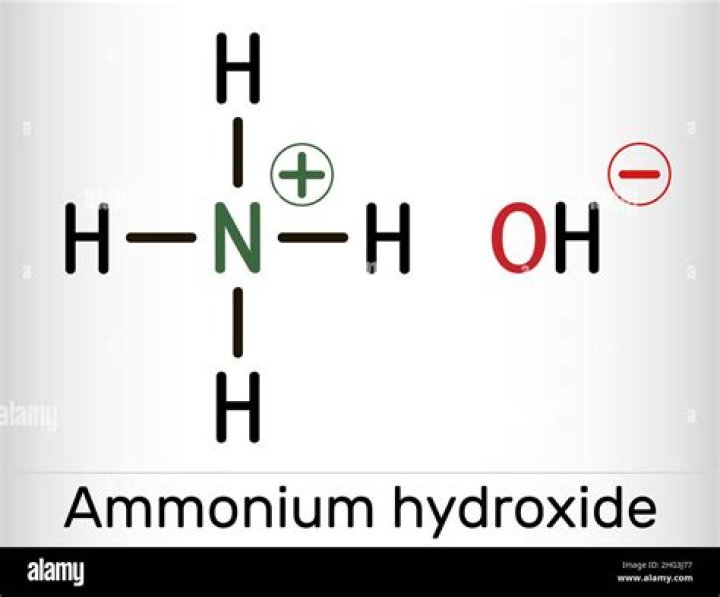
What is the formula for ammonium hydroxide?
Ammonium hydroxide PubChem CID 14923 Structure Find Similar Structures Chemical Safety Laboratory Chemical Safety Summary (LCSS Molecular Formula H5NO or NH4OH Synonyms ammonium hydroxide 1336-21-6 Ammonia aqu



