What is the difference between conformational and constitutional isomers?
Constitutional Isomers: Two molecules with the same composition but different constitution (i.e. – same molecular formula, different IUPAC name for constitution). Conformational isomers are temporarily different shapes of the same molecule and for this reason are not classified as isomers in some textbooks.
What is the difference between constitutional isomers and stereoisomers?
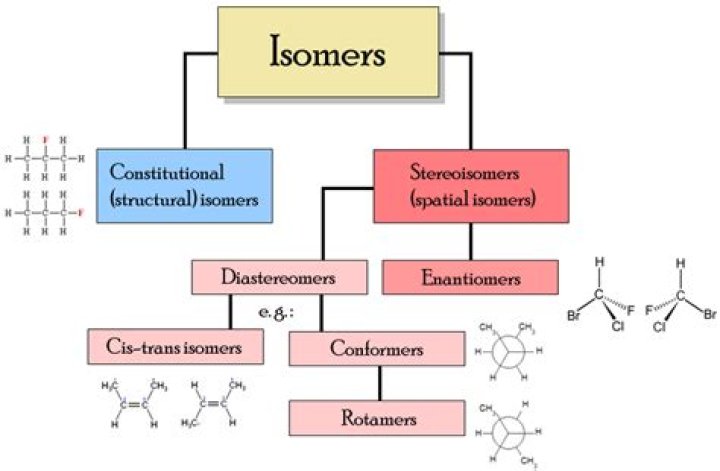
Structural (constitutional) isomers have the same molecular formula but a different bonding arrangement among the atoms. Stereoisomers have identical molecular formulas and arrangements of atoms. They differ from each other only in the spatial orientation of groups in the molecule.
How are conformations different from isomers?
While any two arrangements of atoms in a molecule that differ by rotation about single bonds can be referred to as different conformations, conformations that correspond to local minima on the potential energy surface are specifically called conformational isomers or conformers.
Are stereoisomers constitutional isomers?
The main difference between constitutional isomers and stereoisomers is that constitutional isomers are molecules having the same molecular formula but different atomic arrangements whereas stereoisomers are molecules having the same molecular formula and atomic arrangement but different spatial arrangements.
What is constitutional isomerism?
Constitutional or structural isomers are compounds with the same molecular formula but different structural formulas. eg. 1: Butane and isobutane have the same molecular formula, C4H10, but different structural formulas. Therefore, butane and isobutane are constitutional isomers.
What is meant by constitutional isomers?
What is a cyclic constitutional isomer?
A cyclic molecule that is a constitutional isomer of cyclohexane is: We have to determine the molecular formula of each of the given molecules. Complete answer: We know that the molecules having the same molecular formula but different connectivity of atoms are known as constitutional isomers.
Which is an example of constitutional isomers?
Constitutional isomers have the same molecular formulas, but they have different connectivities. n-Butane and isobutane are examples of constitutional isomers, as are ethanol and dimethyl ether. For example, both ethyl alcohol and dimethyl ether have the same molecular formula: C2H6O.
What are examples of constitutional isomers?
Isomers are defined as compounds that have the same molecular formula, but different structures. There are two ways in which isomers can differ. When they differ in the way the atoms are connected, they are called constitutional isomers. Butane and isobutane, for example, are constitutional isomers.
How do you determine the number of constitutional isomers?
Constitutional isomers are compounds that have the same molecular formula and different connectivity. To determine whether two molecules are constitutional isomers, just count the number of each atom in both molecules and see how the atoms are arranged.
Which compounds have constitutional isomers?
Constitutional isomers have the same molecular formulas, but they have different connectivities. n -Butane and isobutane are examples of constitutional isomers, as are ethanol and dimethyl ether. Constitutional isomers can also have different functional groups.
What is the difference between structural and constitutional isomers?
Structural isomers are isomers that have the same component atoms but they are arranged differently from each other. Structural isomerism is also known as constitutional isomerism. Contrast this with stereoisomerism , where isomers have the same atoms in the same order and with the same bonds, but oriented differently in three-dimensional space.



