What is the difference between neopentane and pentane?
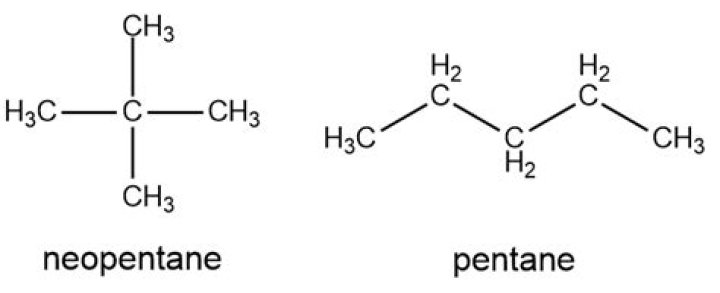
Neopentane is almost spherical, with a small surface area for intermolecular interactions, whereas n-pentane has an extended conformation that enables it to come into close contact with other n-pentane molecules.
What is the structure of neopentane?
C5H12
Neopentane/Formula
Why is neopentane more stable than n-pentane?
pentane is a linear molecule with molecular formula C5H12. It has 3 structural isomers- pentane, isopentane and neopentane. The branched isomers are more stable than pentane because they have lower heat of formation and heat of combustion. hence more branching increases stability.
Are pentane and neopentane isomers?
Isomers (Constitutional) Examples are for butane and isobutane, or for pentane, isopentane, and neopentane. Sometimes referred to as Structural Isomers, Constitutional Isomers have the same mass, but different physical and chemical properties.
What is the meaning of neopentane?
Definition of neopentane : a gaseous or very volatile liquid hydrocarbon (CH3)4C found in small amounts in petroleum and natural gas; dimethyl-propane or tetramethyl-methane.
What is structural formula of pentane?
Pentane/Formula
Which has higher melting point n-pentane or neopentane?
Notice that n-pentane is roughly sausage-shaped, whereas neopentane is a spherical ball. Neopentane molecules pack better into a crystalline lattice, so more energy is required to melt the solid; thus its melting point is the highest of the three isomers.
What is the condensed structural formula for neopentane?
Neopentane
| PubChem CID | 10041 |
|---|---|
| Structure | Find Similar Structures |
| Chemical Safety | Laboratory Chemical Safety Summary (LCSS) Datasheet |
| Molecular Formula | C5H12 |
| Synonyms | NEOPENTANE 2,2-Dimethylpropane Propane, 2,2-dimethyl- 463-82-1 Tetramethylmethane More… |
Which has more boiling point isopentane or neopentane?
The n-pentane melts higher than isopentane because it can assume a rodlike shape in which adjacent molecules can pack closely together. But neopentane melts more than 100° higher than either of its isomers. The boiling points of the three isomers are nearly in the opposite order.
Why does isopentane have a lower melting point than pentane?
The structures of n-pentane and isopentane are: As we can see that branching is present in isopentane. This decreases the surface area of molecule and makes it compact which results in decrease of intermolecular forces between different molecules. Lower the attractive intermolecular forces lower is the melting point.
What is n-pentane isopentane and neopentane?
The term may refer to any of three structural isomers, or to a mixture of them: in the IUPAC nomenclature, however, pentane means exclusively the n-pentane isomer; the other two are called isopentane (methylbutane) and neopentane (dimethylpropane).
Is neopentane Iupac name?
Neopentane, also called 2,2-dimethylpropane, is a double-branched-chain alkane with five carbon atoms….Neopentane.
| Names | |
|---|---|
| Preferred IUPAC name 2,2-Dimethylpropane | |
| Other names Neopentane Tetramethylmethane | |
| Identifiers | |
| CAS Number | 463-82-1 |
What is the difference between neopentane and n-pentane?
Neopentane is almost spherical, with a small surface area for intermolecular interactions, whereas n -pentane has an extended conformation that enables it to come into close contact with other n -pentane molecules. As a result, the boiling point of neopentane (9.5°C) is more than 25°C lower than the boiling point of n -pentane (36.1°C).
How many isomers are there of n-1653 pentane?
There are two isomers of n-pentane: isopentane and neopentane. The word “pentane” usually refers to n-pentane, that is, its straight chain isomer. N-pentane (n-pentane), with the chemical formula of C5H12, is the fifth member of alkane. The boiling point of n-1653 pentane is 36.1 ℃.
What is the chemical formula of neneopentane?
Neopentane, also known as 2,2-dimethylpropane and pentane, has a chemical formula of C5H12. It is a toxic chemical product with a boiling point of 10 ° C. Pentane can be obtained by catalytic cracking and thermal decomposition of natural gas or petroleum.
What is the Lewis structure for pentane?
Pentane Lewis Structure Lewis structures or electron dot structures are diagrams that represent the distribution of valence electrons of atoms within a molecule. Each dot represents an electron, and a pair of dots between chemical symbols for atoms represents a bond.



