What is the enthalpy for Br2?
111.852
Selected ATcT enthalpy of formation based on version 1.118 of the Thermochemical Network
| Species Name | Formula | ΔfH°(298.15 K) |
|---|---|---|
| Bromine atom | Br (g) | 111.852 |
What is the enthalpy of bromine?
The enthalpy of formation for Br (monoatomic gas) is 111.881 kJ/mol.
How do you calculate enthalpy change of atomization?
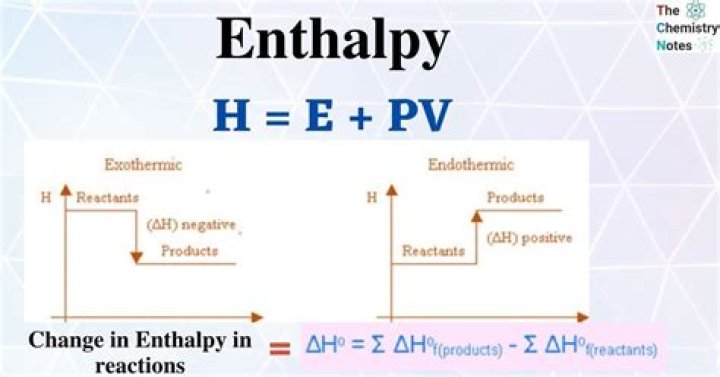
Let us now learn about the calculation of the enthalpy change of atomization. When pressure is held constant, the heat change is equal to the change in the system’s internal energy. Hence, the enthalpy of atomisation is equal to the sum of the total enthalpies of vaporization and fusion.
For which of the following enthalpy of atomisation is zero?
Account for: The enthalpy of atomisation is lowest for Zn in 3d series of the transition elements.
Is Br2 L → Br2 g exothermic?
The Attempt at a Solution The chemical reaction is going from liquid to gas, therefore the entropy is increasing because the reaction is causing more macrostates due to the greater area of distribution for the gas molecules. The reaction is exothermic, because its giving off heat.
What is the standard state of Br2?
liquid
At 298 K and 1 atm, the standard state of Br2 is a liquid, whereas the standard state of I2 is a solid. The enthalpy changes for the formation of Br2(g) and I2(g) from these elemental forms at 298 K and 1 atm are given in the table above.
What is the standard enthalpy of formation of Fe’s )?
| Substance | State | ∆H°f |
|---|---|---|
| Fe | s | 0 |
| Fe(OH)3 | s | −823.0 |
| Fe2O3 | s | −824.2 |
| Fe3O4 | s | −1118.4 |
What is the enthalpy of formation of HBr?
Selected ATcT enthalpy of formation based on version 1.122 of the Thermochemical Network
| Species Name | Formula | ΔfH°(298.15 K) |
|---|---|---|
| Hydrogen bromide | HBr (g) | -35.66 |
What does atomization mean?
Definition of atomize transitive verb. 1 : to treat as made up of many discrete units. 2 : to reduce to minute particles or to a fine spray. 3 : divide, fragment an atomized society also : to deprive of meaningful ties to others atomized individuals.
What is enthalpy of atomization give an example?
Enthalpy of atomization, ΔaH0, is the change in enthalpy when one mole of bonds is completely broken to obtain atoms in the gas phase. For example: atomization of methane molecule. For diatomic molecules, enthalpy of atomization is equal to the enthalpy of bond dissociation.
What is the meaning of atomisation?
Atomization is the process of converting an analyte in solid, liquid or solution form to a free gaseous atom. It is the transformation of a bulk liquid into a spray of liquid droplets in a surrounding gas or vacuum.
What is meant by enthalpy of atomisation?
Enthalpy of atomization is the amount of enthalpy change when a compound’s bonds are broken and the component atoms are separated into individual atoms.
What is the change in enthalpy of atomization?
This change in enthalpy can be due to the enthalpy of atomization, solution etc. Some common enthalpy changes are explained below: Enthalpy of atomization, Δ a H 0, is the change in enthalpy when one mole of bonds is completely broken to obtain atoms in the gas phase.
What is the atomic number of bromine?
Bromine – Periodic Table Element Bromine Atomic Number 35 Symbol Br Element Category Halogen Phase at STP Liquid
What is the latent heat of vaporization of bromine?
Latent heat of vaporization of Bromine is 15.438 kJ/mol. In case of liquid to gas phase change, this amount of energy is known as the enthalpy of vaporization, (symbol ∆Hvap; unit: J) also known as the (latent) heat of vaporization or heat of evaporation.
What is the enthalpy of fusion and vaporization of BR?
Enthalpies Enthalpy of fusion: 5.8 (per mol Br atoms) kJ mol-1 Enthalpy of vaporisation: 14.8 (per mole Br atoms) kJ mol-1 Enthalpy of atomisation: 112 kJ mol-1



