What is the formula of isobutane?
C4H10
Isobutane/Formula
What is n-butane and isobutane?
Isobutane, butane and propane are different chemical molecules with different physical properties. Butane (n-butane) C4H10, has four carbon atoms in a straight chain with 10 hydrogen atoms, while Isobutane (i-butane) is an isomer of butane with a branched structure. Propane C3H8 is an entirely different chemical gas.
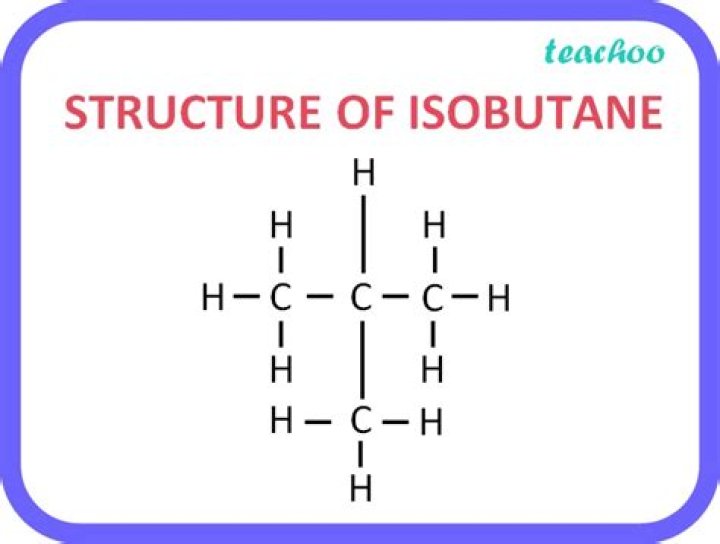
What is the structure of n-butane and isobutane?
two molecules, called butane and isobutane, are constitutional isomers. They are different molecules with different chemical and physical properties. Butane has its four carbon atoms bonded in a continuous chain. Isobutane has a branched structure.
How do you convert n-butane to isobutane?
A method for converting n-butane into isobutane through using a gold supported molecular sieve catalyst belongs to the technical field of catalysts. High-dispersion nano-gold particles are formed through the strong reaction of gold and high-silicon zeolite when nano-gold is loaded on the high-silicon zeolite.
What is butane isomerization?
Normal butane is converted into isobutane in a process called isomerization. Butane isomerization happens in an isomerization (“Isom”) unit, also called a butamer unit. The butamer process converts the straight chain molecule into the branched molecule, also called the “isomer” of normal butane.
Are n butane and isobutane structural isomers?
In the case of n-butane, all carbon atoms are in straight-chain whereas, in the case of isobutane, there is a side chain in the molecule. So they have different connectivity of atoms and are constitutional isomers of each other.
What is N in N butane?
Isobutane can be represented as i-butane and normal butane can be represented as n-butane. The “n” in the front is letting you know that you are talking about normal butane which is linear and the butane most (99%) of people are referring to when talking about butane.
What is N in n-butane?
Does butane and isobutane have same formula?
Butane is unbranched, and isobutane is branched. 2. Both have the same molecular formula, but the structural formula is different. Butane has four carbon atoms in the straight chain, whereas isobutane has only three carbon atoms in the straight chain.
How will you convert propane to propene?
Step 1 : First convert the propane to 1-chloropropane by in presence of sunlight . The chlorine react with the alpha carbon. Step 2 : Now 1-chloropropane react with alcoholic potassium hydroxide to give propene as a product by loss of HCl.
What is the product formed when n-butane is isomerized?
For isomerization of n-butane in the gas phase, the reaction proceeds with the dehydrogenation of n-butane to form a linear alkene (butene) intermediate. This reaction is catalyzed by the metallic function. II. Butene is then protonated to a carbenium ion, which subsequently is isomerized to a branched carbenium ion.
What is the difference between n butane and isobutane?
Difference between n butane and isobutane is that isobutane (i-butane) is an isomer of normal butane (n-butane). That means it has the same chemical formula as butane – C4H10 – but has a different arrangement of its atoms, as you can see in the 3-D model images. (Isobutane molecule model shown)
What is the molecular formula for butane?
Butane PubChem CID 7843 Structure Find Similar Structures Chemical Safety Laboratory Chemical Safety Summary (LCSS Molecular Formula C4H10 or CH3CH2CH2CH3 Synonyms BUTANE n-Butane 106-97-8 Diethyl
What are the structural isomers of butanes?
The term butanes includes the two structural isomers, n-butane [106-97-8], CH3CH2CH2CH3, and isobutane , (CH3)2CHCH3 (2-methylpropane). Eastman Ad, Mears D; Hydrocarbons, C1-C6. Kirk-Othmer Encyclopedia of Chemical Technology (1999-2013).
Is butane organic or inorganic?
Butane is an organic compound. This molecule can be found in several forms known as isomers. Isobutane is a structural isomer of butane. Both butane and isobutane are gaseous hydrocarbon compounds. They are hydrocarbons because these compounds are composed only of C and H atoms.



