What is the hybridization of PBr5?
The expected hybridization of phosphorus in PBr5 P B r 5 is sp3d s p 3 d . The sp3d s p 3 d hybridization is observed for atoms which…
What shape is PBr5?
PBr5 ‘s molecular geometry is trigonal bipyramidal. Let’s start with its Lewis structure. P has 5 valence electrons and Br has 7 valence electrons, which means that a total of 5+5⋅7=40 electrons must be accounted for.
Does PBr5 exist?
PBr5 is exists as PBr4+ Br- phosphorus with 4 bromines with a +ve charge, with bromide as the counter ion and it completely dissociates to PBr3 and Br2 upon heating. Similarly, Pl5 exist as [PI4] -I.
What is the hybridization of PCL 6?
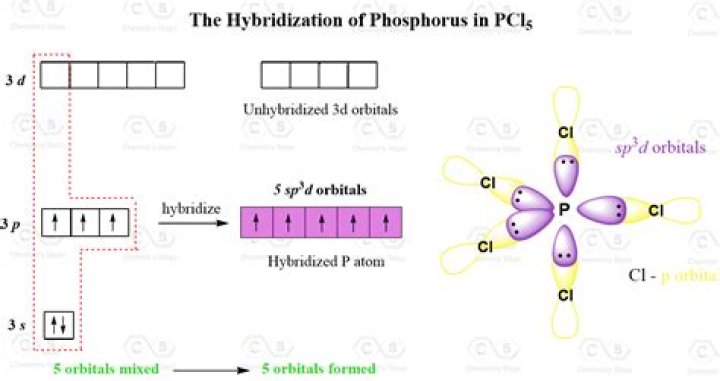
PCl6- has octahedral structure and sp3d2 hybridization for its central atom whears the PCl4+ has tetrahedral structure and sp3 hybridization. PCl5 in gas form has disphenoid structure with sp3d hybridization.
What is hybridisation of central atom of anionic part of PBr5 in crystalline state?
In solid state PBr5 exist as [PBr4]+Br-. Anion part is Bromide ion. It has four valence electrons which are bonded with four bromide ions. So, the hybridisation of central atom in cation part is sp3.
How does PBr5 exist in solid state?
PBr5 in solid state exists as [PBr4 ]^+Br^- . The expected shape of PBr4^+ is(1) Tetrahedral (2) Trigonal bipyramidal(3) Square planar (4) Octahedral.
How many electron groups are around the central phosphorus atom of PBr5?
Phosphorus pentafluoride has 5 regions of electron density around the central phosphorus atom (5 bonds, no lone pairs). The resulting shape is a trigonal bipyramidal in which three fluorine atoms occupy equatorial and two occupy axial positions.
Why is PBr5 unstable?
PCl5 Exit becaue size of chlorine is comparable to sulphur so they easily form bonds which are stable. But due to greater size of Br and I they cannot form stable bonds with the sulphur. So PBr5 and PI5 does not exist.
What is hybridization of so2?
In sulphur dioxide, the hybridization that takes place is sp2 type. To determine this, we will first look at the sulphur atom which will be the central atom. During the formation of SO2, this central atom is bonded with two oxygen atoms and their structure can be represented as O=S=O.
What is hybridization of co2?
Carbon dioxide has an sp hybridization type. This hybridization type occurs as a result of carbon being bound to the other two atoms. Bonds can be either one single + one triple bond or two double bonds.
Is PCl5 an ionic halide?
Yes! Lattice energy is usually the largest factor in determining the stability of an ionic solid. The extra energy gained by the lattice energy more than compensates for the energy needed to transfer a chloride ion from one PCl5 molecule to another. Thus, PCl5 exists as an ionic solid.
What is the molecular geometry of PBr5?
1 Answer. PBr5 ‘s molecular geometry is trigonal bipyramidal. Let’s start with its Lewis structure. P has 5 valence electrons and Br has 7 valence electrons, which means that a total of 5+5⋅7 = 40 electrons must be accounted for. P ‘s bonds with the five Br atoms account for 5⋅2 = 10 electrons, while the three lone pairs on each Br atom complete…
What are the types of hybridization?
Hybridization is the mixing of two non equivalent atomic orbitals. The result of hybridization is the hybrid orbital. There are many types of hybrid orbitals formed by mixing s, p and d orbitals. The most common hybrid orbitals are sp3, sp2 and sp.
What is the importance of hybridization?
Hybridization is the one of the important application of agriculture .Hybridization is the process of crossing two genetically different individuals to create new genotypes.
What is the process of hybridization?
Hybridization is the process of interbreeding between individuals of different species (interspecific hybridization) or genetically divergent individuals from the same species (intraspecific hybridization).



