What is the molecular geometry of PCl3?
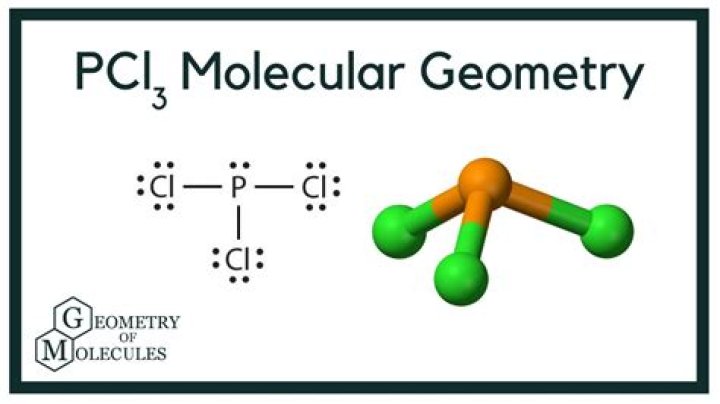
Trigonal pyramidal
PCl3
| Central atom: | P |
|---|---|
| 3 x Cl contibute: | 3 e− |
| Total VSE: | 8 |
| Total VSEP: | 4 |
| Geometry: | Trigonal pyramidal (based on tetrahedral) |
What is the molecular geometry for dihydrogen monoxide?
Dihydrogen monoxides creates a bent shape when following the priciples of VSEPR. It is also polar due to the partially negative oxygen and positive hydrogen.
Is PCl3 At shaped molecule?
The shape of PCl3 molecule is A trigonal bipyramid class 11 chemistry JEE_Main.
What is the formula for carbon monoxide?
CO
Carbon monoxide/Formula
Is PCl3 planar or nonplanar?
PCl3 is a trigonal planar shape. The vector addition of the 3 dipole moments gives a better of 0. Hence its non-polar.
Is PCl3 an ionic compound?
Phosphorus trichloride
Phosphorus trichloride/IUPAC ID
What is the molecular geometry of the compound PCl3?
PCl3 Molecular Electron Geometry, Lewis Structure, Bond Angles and Hybridization Name of molecule Phosphorus trichloride ( PCl3) No of Valence Electrons in the molecule 26 Hybridization of PCl3 sp3 hybridization Bond Angles Less than 109 degrees Molecular Geometry of PCl3 Trigonal Pyramidal
What is the hybridization of PCl3?
So as four hybrid orbitals are formed, the hybridization of PCl3 is sp3. PCl3 Molecular Geometry Once you know the molecule’s electron geometry, it is relatively easy to guess the molecular geometry. Here the molecular geometry of Phosphorus Trichloride is trigonal pyramidal.
What is the molecular geometry of phosphorus trichloride?
Here the molecular geometry of Phosphorus Trichloride is trigonal pyramidal. As per the molecular geometry of the molecule, the bond angle of PCl3 should be 109 degrees. But as there is one lone pair of electrons on the central phosphorus atom, the bond angle will reduce from 109 degrees because of the repulsive forces of the lone pair.
How many valence electrons does PCl3 (phosphorus trichloride) have?
Phosphorus Trichloride (PCl3) has a total of 26 valence electrons. PCl3 Lewis Structure Now that we know the total number of valence electrons for Phosphorus Trichloride, we will start drawing the Lewis Dot Structure for this molecule.



