What is the oxidation number of K in KClO?
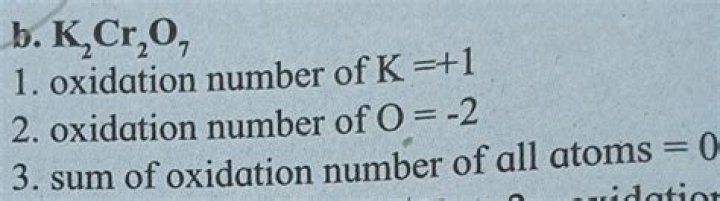
+1
– Potassium $\left( \text{K} \right)$ belongs to group 1 as it is an alkaline metal. The oxidation state of potassium is +1.
Which element is oxidized in KClO?
Similarly, the oxidation state of Cl in KCI is also calculated. According to the oxidation states calculated in Step 1, oxidation state of oxygen changes from -2 to 0 from reactants to products. The element which is oxidized is oxygen.
What are the oxidation numbers of K Cl and O in KClO?
1 Expert Answer Cl can have variable ONs so we must figure it out. Answer: K = +1, Cl = +5, O = -2 (Choice B)
What is the charge of KClO?
Since the entire compound KClO3 has a charge of 0 and the K will have a charge of +1 , the ClO3 must balance the K ‘s +1 in the form of −1 for a net charge of 0 .
In which compound does chlorine have the highest oxidation number KClO?
HClO4
The correct answer is HClO4. Chlorine shows its highest oxidation state in HClO4.
What is the oxidation state of each element in MN cro4 2?
Manganese has positive 4, chromium has positive 6 and oxygen, a negative 2.
Which element is oxidized in the reaction?
The element whose formal oxidation number increases during a reaction is said to have been oxidized . The element whose formal oxidation number decreases during a reaction is said to have been reduced .
In which of the following chlorine has 1 oxidation number?
In case of Cl2O chlorine shows +1 oxidation state.
How do you calculate oxidation number?
The oxidation number of each atom can be calculated by subtracting the sum of lone pairs and electrons it gains from bonds from the number of valence electrons. Bonds between atoms of the same element (homonuclear bonds) are always divided equally.
How to find oxidation number?
Any free element has an oxidation number equal to zero.
How to assign oxidation numbers?
The convention is that the cation is written first in a formula,followed by the anion. For example,in NaH,the H is H-; in HCl,the H is H+.
What is the oxidation number of Cl in KClO3?
number is –1 (as in KCl). To find the oxidation number of Cl in KClO 3 , add the known oxidation numbers and set the sum equal to 0: K + Cl + O 3 = 0 (+1) + x + (3 x (–2)) = 0 x = Cl = +5 The oxidation number of chlorine changes from +5 to –1. 4 125.



