What is the product of bromination of trans stilbene?
Bromination of trans-stilbene produces predominantly meso-1,2-dibromo-1,2-diphenylethane (sometimes called meso-stilbene dibromide), in line with a mechanism involving a cyclic bromonium ion intermediate of a typical electrophilic bromine addition reaction; cis-stilbene yields a racemic mixture of the two enantiomers …
What is the purpose of bromination of trans stilbene?
AIM: The aim of this experiment is to carry out an addition reaction using trans-stilbene and bromine reagent to produce 1,2-dibromo-1,2-diphenylethane.
How do you distinguish CIS stilbene and trans stilbene by UV spectroscopy?
The key difference between cis and trans stilbene is that in cis stilbene, the two phenyl groups are on the same side of the double bond whereas, in trans stilbene, the two phenyl groups are on the opposite sides of the double bond.
Which reagent are used to bromination of trans-stilbene reaction?
You will carry out the bromination reactions of both cis- and trans-stilbene, and from the melting points of the products, you will be able to determine whether the racemic mixture or the meso compound is formed. In these two bromination reactions, pyridinium tribromide will be used as the source of bromine.
Why is the melting point of Z stilbene so much lower than e stilbene?
(Z)-Stilbene is sterically hindered and less stable because the steric interactions force the aromatic rings 43° out-of-plane and prevent conjugation. (Z)-Stilbene has a melting point of 5–6 °C (41–43 °F), while (E)-stilbene melts around 125 °C (257 °F), illustrating that the two compounds are quite different.
Is cis or trans stilbene more stable?
Therefore, generally speaking, the trans (E) isomers are more stable and lower in energy than the cis (Z) isomers.
Why is pyridinium tribromide a better reagent than bromine?
In these two bromination reactions, pyridinium tribromide will be used as the source of bromine. This solid reagent is safer and easier to handle than elemental bromine, which is a highly corrosive liquid.
How do you brominate trans-stilbene?
Bromination of trans-stilbene Weigh 0.4 g of trans-stilbene and place it in an 18 x 150 mm test tube. Add 4 mL of glacial acetic acid and heat the test tube in a water bath to dissolve the solid. Stir with a glass rod until it completely dissolves.
What type of reaction is trans-stilbene and bromine?
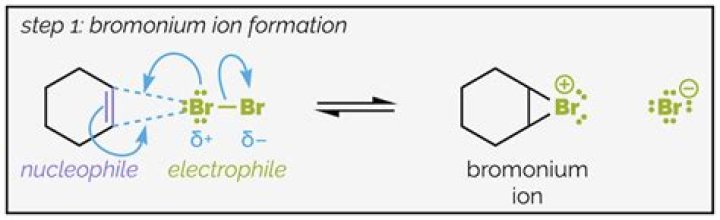
Hence, an addition reaction takes place an 1,2-dibromo-1,2-diphenylethane is formed. MECHANSIM: The alkene, trans-stilbene, acts as a nucleophile and the bromine acts as an electrophile. Hence, the nucleophilic double bond undergoes an electrophilic addition reaction by the bromine reagent.
How do you convert trans-stilbene to diphenylethane?
This practical involves the bromination of trans-stilbene using a 10% bromine dichloromethane solution. Hence, an addition reaction takes place an 1,2-dibromo-1,2-diphenylethane is formed. MECHANSIM: The alkene, trans-stilbene, acts as a nucleophile and the bromine acts as an electrophile.
How do you convert bromine to trans dibromide?
The addition of bromine begins at one side of the double bond (either side is equally likely, but only one option is drawn) and is followed by attack of bromide ion on the bromonium ion (again, attack could occur at either carbon since the ion is symmetric, but only one option is drawn). The result is a trans dibromide, as shown above.



