What is the relationship between orbitals and sublevels?
A sublevel is further divided into orbitals. In an atom, the region of space which has the highest probability of electron is called an orbital. In the case of a hydrogen atom, 99 per cent of the time the electron is found surrounding the nucleus somewhere within a spherical region.
What is the relationship between orbitals and electrons?
Electrons that occur together in an orbital are called an electron pair. An electron will always try to enter the orbital with the lowest energy. An electron can occupy an orbital on its own, but it would rather occupy a lower-energy orbital with another electron before occupying a higher-energy orbital.
How many orbitals are in each sublevel?
Recall that the four different sublevels each consist of a different number of orbitals. The s sublevel has one orbital, the p sublevel has three orbitals, the d sublevel has five orbitals, and the f sublevel has seven orbitals. In the first period, only the 1s sublevel is being filled.
What is the relationship between the energy sublevels and principal quantum numbers?
Each principal energy level may be split into closely spaced sublevels (or sublevels) as specified by l. This quantum number may be more aptly named the orbital shape quantum number, since each orbital in a given type of sublevel (i.e., a given value of l) has the same “electron cloud” shape.
What is the relationship between N and the total number of orbitals in a main energy level?
All orbitals that have the same value of n are said to be in the same shell (level). For a hydrogen atom with n=1, the electron is in its ground state; if the electron is in the n=2 orbital, it is in an excited state. The total number of orbitals for a given n value is n2.
In what order do the levels get filled with electrons?
In order as: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p… 1s will be filled first, with the maximum of 2 electrons. 2s will be filled next, with the maximum of 2 electrons. 2p will be filled next, with the maximum of 6 electrons.
What is the difference between the electron orbital and electron shells?
The main difference between shell subshell and orbital is that shells are composed of electrons that share the same principal quantum number and subshells are composed of electrons that share the same angular momentum quantum number whereas orbitals are composed of electrons that are in the same energy level but have …
What is the difference between Orbit and orbitals?
Differences between Orbit and Orbitals An orbit is the simple planar representation of an electron. An orbital refers to the dimensional motion of an electron around the nucleus in a three-dimensional motion. An orbital can simply be defined as the space or the region where the electron is likely to be found the most.
What is the relationship between the electron configuration of an element and the period?
the number of circles in the electronic configuration of an element is represented in the periodic table as the period number that element is situated in. the number of electrons in the outermost shell of an element is represented in the periodic table as the group number that element is situated in.
What atomic orbitals are related to AP sublevel?
So, in order to have a p-sublevel, you need to have n>1 , since the first energy level only has an s-sublevel. Therefore, the p-sublevel has a total of three p-orbitals: px , py , and pz .
What is the difference between principal energy levels orbitals and sublevels?
Orbitals of equivalent energy are grouped in sublevels. Each orbital can contain a maximum of two electrons. The first principal energy level contains only an s sublevel; therefore, it can hold a maximum of two electrons. Each principal energy level above the first contains one s orbital and three p orbitals.
What is the difference between orbitals and sublevels?
The sublevels contain orbitals. Orbitals are spaces that have a high probability of containing an electron. In other words, an orbital is an area where the electrons live. There can be two electronsin one orbital maximum. The s sublevel has just one orbital, so can contain 2 electrons max.
What is the relationship between electron orbitals and energy?
The less the distance between the electron and nucleus, the lower is the electron’s energy. The 3s, 4s, and 5s orbitals gradually move farther from the nucleus. Summary: A sublevel is divided into orbitals.
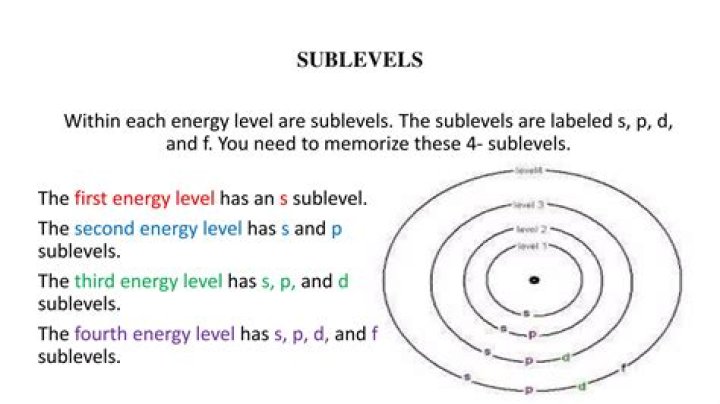
What are the sublevels of electron configuration level 4?
Level 4 has 4 sublevels – s, p, d, and f. These are pictured below. The sublevels contain orbitals. Orbitals are spaces that have a high probability of containing an electron. In other words, an orbital is an area where the electrons live. There can be two electronsin one orbital maximum.
What is the maximum number of electrons in the p-sublevel?
The p sublevel has 3 orbitals, so can contain 6 electrons max. The d sublevel has 5 orbitals, so can contain 10 electrons max. And the 4 sublevel has 7 orbitals, so can contain 14 electrons max. In the picture below, the orbitals are represented by the boxes.



