What is the relationship between rate and concentration for a second order reaction?
Second-Order Reactions For a second-order reaction, t 1 / 2 t 1 / 2 is inversely proportional to the concentration of the reactant, and the half-life increases as the reaction proceeds because the concentration of reactant decreases.
How does a graph indicate second order?
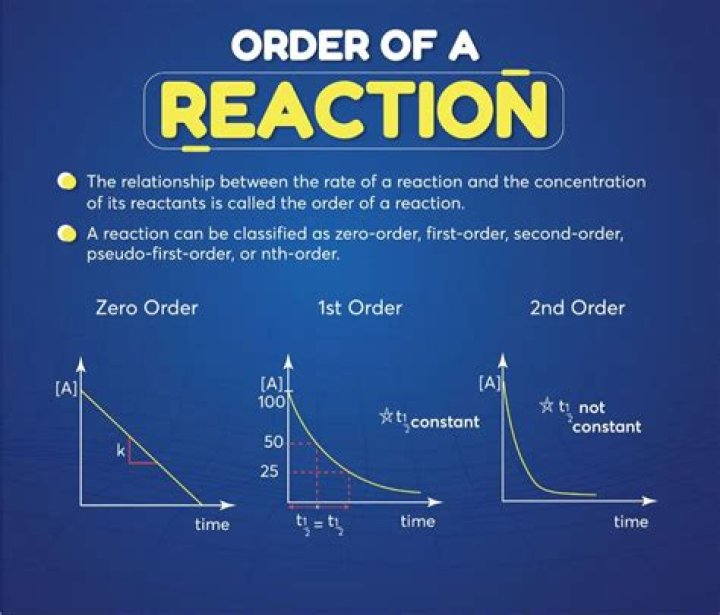
If you get a straight line with a negative slope, then that would be first order. For second order, if you graph the inverse of the concentration A versus time, you get a positive straight line with a positive slope, then you know it’s second order.
How do you find the order of reactions from concentration and time?
Take three consecutive points from the concentration versus time data. Calculate ΔyΔx for the first and second points. The concentration is the y value, while time is the x value. Do the same for the second and third point.
How do you find the order of reactions with concentration and time?
How do you know if a reaction is first or second order?
Add the exponents of each reactant to find the overall reaction order. This number is usually less than or equal to two. For example, if reactant one is first order (an exponent of 1) and reactant two is first order (an exponent of 1) then the overall reaction would be a second order reaction.
How can you use a graph of reactant concentration versus time to determine the rate of a chemical reaction?
For a first-order reaction, a plot of the natural logarithm of the concentration of a reactant versus time is a straight line with a slope of −k. For a second-order reaction, a plot of the inverse of the concentration of a reactant versus time is a straight line with a slope of k.
How do you know if its first or second order?
Initial Rate (M/s) Determine the reaction order and the rate constant. If a plot of reactant concentration versus time is not linear but a plot of 1/reaction concentration versus time is linear, then the reaction is second order.
How do you find the second order of concentration?
For second order, if you graph the inverse of the concentration A versus time, you get a positive straight line with a positive slope, then you know it’s second order. The other thing; is you graph 3 or 4 points, and you notice they’re straight, then that would be plenty to help you figure out. Two points not enough.
What is the graph of concentration as a function of time?
Conversely, a graph of the concentration of any product as a function of time is a straight line with a slope of (k), a positive value. The graph of a zeroth-order reaction. The change in concentration of reactant and product with time produces a straight line.
What is a first order graph?
First order, would be natural log of concentration A versus time. If you get a straight line with a negative slope, then that would be first order. For second order, if you graph the inverse of the concentration A versus time, you get a positive straight line with a positive slope, then you know it’s second order.
How do you determine the Order of a reaction from a graph?
Determining Order of a Reaction Using a Graph – Concept. First order, would be natural log of concentration A versus time. If you get a straight line with a negative slope, then that would be first order. For second order, if you graph the inverse of the concentration A versus time, you get a positive straight line with a positive slope,…



