What is the strongest intermolecular force in c4h10?
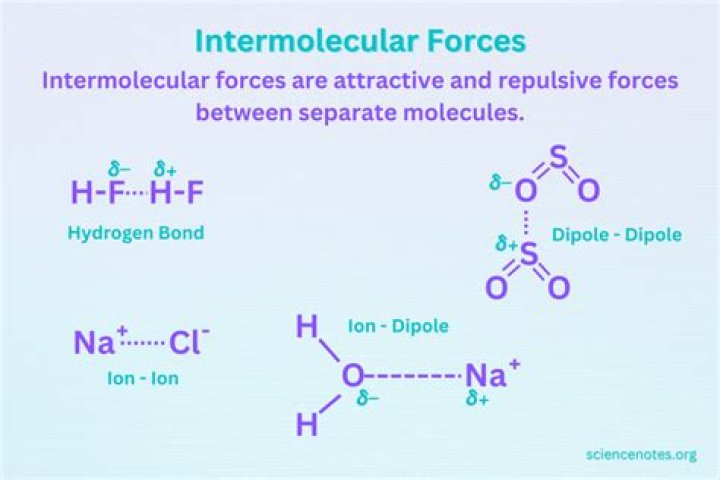
There are three non-ionic forces. The first and the strongest is hydrogen bonding which occurs between a H atom and either a N, O, or F, causing a very strong attractive force between the positive H and electronegative atom.
What is the strongest intermolecular force in N2?
1 Expert Answer In H2O, the intermolecular forces are not only hydrogen bonging, but you also have dipole-dipole and dispersion forces. In N2, you have only dispersion forces.
Does N2 have weak intermolecular forces?
N2 has very weak intermolecular forces which make it a gas. Nitrogen gas (N2) is diatomic linear and non-polar because both nitrogen atoms have the same degree of electronegativity and they cancel each other. However, weak London dispersion forces do exist by means of the creation of temporary dipoles.
What is the strongest intermolecular force in no2?
Hydrogen bonds
Correct answer: This type of intermolecular force is the attraction that occurs between hydrogen atoms and the lone pairs on atoms of oxygen, nitrogen and/or fluorine. Hydrogen bonds are the strongest while dispersion forces are the weakest.
What is the intermolecular forces of Ch3oh?
Ch3oh intermolecular forces has hydrogen bonding, dipole dipole attraction and London dispersion forces.
What type of interparticle forces holds liquid n2 together?
Dipole–Dipole Interactions.
What is the intermolecular forces of NH3?
You know that, ammonia is a polar molecules. it exhibits, dipole-dipole intraction, induced attraction, and London dispersion forces. NH3 is called dipole dipole because nh3 make N-H bond, it directly make hydrogen bonding. hydrogen is bound to nitrogen and it make hydrogen bonds properly.
Which molecules have the strongest intermolecular forces?
The answer is hydrogen-bonding, the which occurs when hydrogen is bound to a strongly electronegative element such as oxygen, or nitrogen, or fluorine. The hydrides of all these elements have disproportionately high boiling points.
Which compound has the strongest intermolecular forces?
HF (boiling point = 19.4 degrees Celsius) has the strongest intermolecular forces.
What intermolecular forces does NO2 have?
NO2 is a polar molecule (due to it having a bent shape) so the most significant intermolecular forces would be dipole-dipole forces.
What’s the strongest intermolecular force in CH3OH?
yes, hydrogen bond is 10 time stronger then all dipole -dipole forces. therefore, hydrogen bond is more stronger then dipole-dipole forces.
What intermolecular forces are present in C4H10?
What intermolecular forces are present in c4h10? 1 C6H14: Dispersion forces only. 2 C4H10: Dispersion forces only. 3 C2H5OH: Dispersion forces, dipole-dipole forces, and hydrogen bonding. 4 and. 5 HF: Dispersion forces, dipole-dipole forces, and hydrogen bonding. 6 C6H14 and C4H10 are nonpolar organic compound as there are non polar carbon chain.
What is the difference between C4H10 and C6H14?
C4H10: Dispersion forces only. C2H5OH: Dispersion forces, dipole-dipole forces, and hydrogen bonding. HF: Dispersion forces, dipole-dipole forces, and hydrogen bonding. C6H14 and C4H10 are nonpolar organic compound as there are non polar carbon chain.
What type of intermolecular attraction has the strongest force?
The hydrogen bonds are the strongest force, but the other types of intermolecular attraction are still present. Dipole-Dipole Interactions.
What type of intermolecular forces exist between N2 molecules?
Hence, they are termed as London forces. So the type of intermolecular force that exists is dipole-dipole forces. Because N2 molecules are nonpolar, the intermolecular forces between them are dispersion forces, also called London forces.



