What is the transition state of transition state theory?
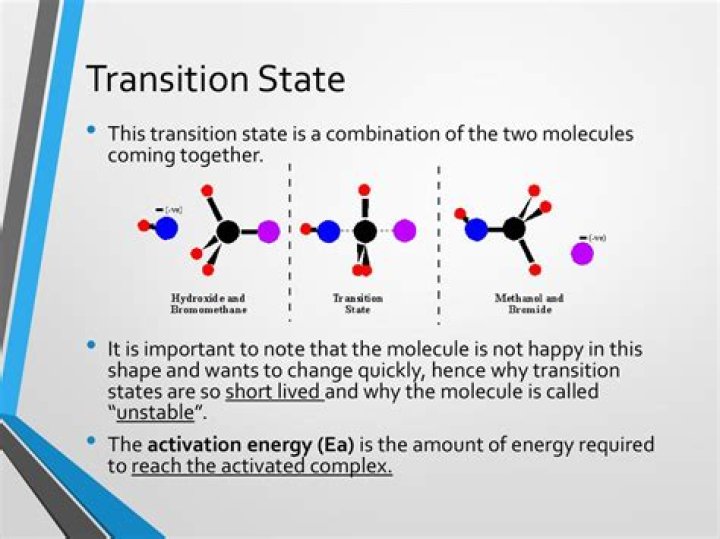
According to transition state theory, between the state in which molecules exist as reactants and the state in which they exist as products, there is an intermediate state known as the transition state. The species that forms during the transition state is a higher-energy species known as the activated complex.
What is the purpose of transition state theory?
Transition state theory (TST) explains the reaction rates of elementary chemical reactions. The theory assumes a special type of chemical equilibrium (quasi-equilibrium) between reactants and activated transition state complexes. TST is used primarily to understand qualitatively how chemical reactions take place.
What is meant by transition state?
The transition state of a chemical reaction is a particular configuration along the reaction coordinate. It is defined as the state corresponding to the highest potential energy along this reaction coordinate. It is often marked with the double dagger ‡ symbol.
What happens in the transition state?
The transition state is a high-energy state, and some amount of energy – the activation energy – must be added in order for the molecule reach it. Because the transition state is unstable, reactant molecules don’t stay there long, but quickly proceed to the next step of the chemical reaction.
What is transition state theory related to Mcq?
Transition state theory postulates that the reacting molecules form unstable intermediates, which are subsequently decomposed to products. Explanation: Transition state theory proposes that reactants form intermediates called activated complex, which then stabilizes by product formation.
Who developed the transition state theory?
The theory was formulated by Erying and Polanyi in 1935 (The calculation of absolute reaction rates [4]) in order to explain bimolecular reactions based on the relationship between kinetics and thermodynamics [5, 6].
Which is incorrect about transition state theory?
Which of the following is incorrect about Transition state theory? Explanation: The activated complex formation step is the fastest. The rate determining step is the slowest of all the steps involved in a reaction. Hence, the decomposition of activated complex is the rate determining step.
What is transition state in biochemistry?
By definition, the transition state is the transitory of molecular structure in which the molecule is no longer a substrate but not yet a product. All chemical reactions must go through the transition state to form a product from a substrate molecule.
What is the definition of a transition state?
The transition state of a chemical reaction is a particular configuration along the reaction coordinate. It is defined as the state corresponding to the highest potential energy along this reaction coordinate.
What is the difference between intermediate and transition state?
Whereas, transition state is just the state before formation of a new molecule. An intermediate differs from a transition state in that the intermediate has a discrete lifetime. A transition state last for just one bond vibration cycle. Intermediates may be highly stable or unstable molecules.
What are transition states in chemistry?
Transition state. The transition state of a chemical reaction is a particular configuration along the reaction coordinate. It is defined as the state corresponding to the highest energy along this reaction coordinate.
What is transition state energy?
The transition state’s energy or, in terms of a reaction, the activation energy is the minimum energy that is needed to break certain bonds of the reactants so as to turn them into products.



