What is used as a titrant in hardness determination?
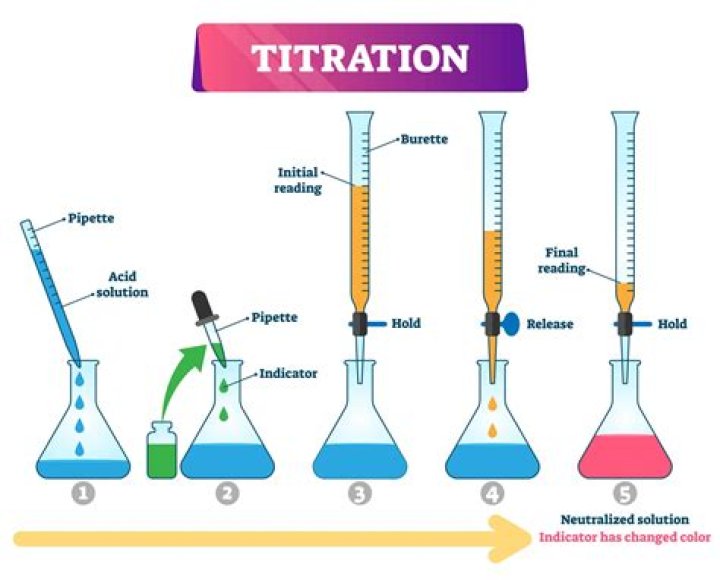
Hardness of water is determined by titrating with a standard solution of ethylene diamine tetra acetic acid (EDTA) which is a complexing agent. Since EDTA is insoluble in water, the disodium salt of EDTA is taken for this experiment. EDTA can form four or six coordination bonds with a metal ion.
What is hardness titration?
1.0 Background. Hardness of water is a measure of the total concentration of the calcium and magnesium ions expressed as calcium carbonate. In this procedure, a water sample is buffered to pH 10.1 and indicator is then added to the buffered sample.
Why CACO3 is used in hardness?
For water hardness levels, we measure parts per million of minerals including calcium carbonate (CACO3) in the water. Calcium carbonate is the compound in hard water that causes limescale build-up. Soft water typically has less than 50 ppm of calcium carbonate. Hard water has over 200ppm.
Which indicator is used in hardness determination?
Eriochrome Black T is a complexometric indicator that is used in complexometric titrations, e.g. in the water hardness determination process. It is an azo dye.
What are the reagents used in the estimation of hardness?
Oxalic acid. EDTA. Sodium citrate.
Why is EDTA an appropriate titrant for the determination of water hardness?
In the determination of water hardness, ethylene- diaminetetraacetic acid (EDTA) is used as the titrant that complexes Ca2+ and Mg2+ ions. This color change signals the end point, as it takes place when the EDTA, after complexing all of the unbound Ca2+ and Mg2+ ions, removes the Mg2+ ion bound to the indicator.
How do you measure water hardness in a titration?
Water hardness can be measured using a titration with ethylenediaminetetraacetic acid (EDTA). The ionised form of EDTA is shown on the right. EDTA dissolved in water forms a colourless solution. An indicator, known as a metal ion indicator, is required for the titration.
How do you determine hardness?
Water hardness can be easily measured using a simple soap test kit that will measure in “grains of hardness” (a little bottle with a line marked on it which you fill to the line with water, add a drop of soap, and shake to look for suds. More drops of soap – more degrees of hardness).
Why is alkalinity expressed as CaCO3?
Why is alkalinity reported as “mg/L as CaCO3”? Units of mg/L are a “mass dissolved in a liquid.” Reporting alkalinity as “mg/L as CaCO3” specifies that the sample has an alkalinity equal to that of a solution with a certain amount of calcium carbonate (CaCO3) dissolved in water.
What is carbonate and Noncarbonate hardness?
Carbonate and Non-carbonate Hardness Carbonate hardness is primarily caused by the carbonate and bicarbonate salts of calcium and. magnesium. Non-carbonate hardness is a measure of calcium and magnesium salts other than. carbonate and bicarbonate salts (such as calcium sulfate, CaSO4, or magnesium chloride, MgCl2).
Why we use EDTA in complexometric titration?
The most common indicators in complexometric titrations are organic dyes which function by forming a colored complex with the metal ion being titrated. During the reaction, EDTA replaces the indicator to form a more stable complex with metal and when the reaction is completed the change for the color is observed.
What is Clark’s process?
Cold lime softening, also known as Clark’s process, is used to reduce raw water hardness, alkalinity, silica, and other constituents. This helps prepare water for direct use as cooling tower makeup or as a first-stage treatment followed by ion exchange for boiler makeup or RO Reject recycle.



