When an electron cloud is easily distorted?
The ease with which a particleís electron cloud can be distorted is called its polarizability.
What is distortion of electron cloud?
Polarization of an anion is distortion of its electron cloud by an adjacently placed cation.
How would you describe an electron cloud?
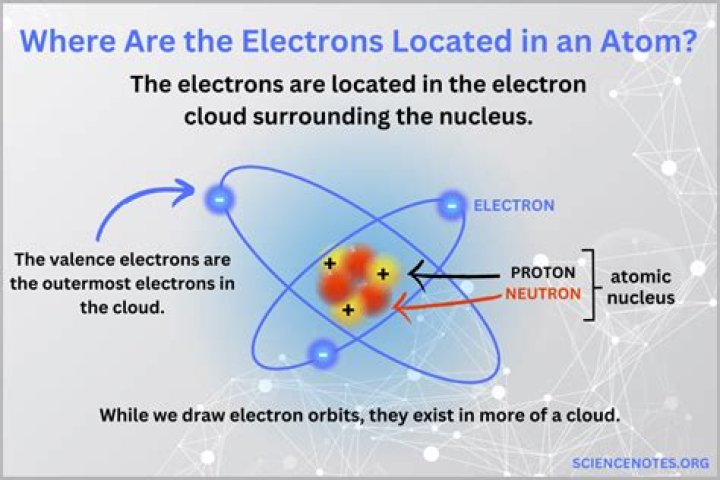
An electron cloud is the region of negative charge surrounding an atomic nucleus that is associated with an atomic orbital. It is defined mathematically, describing a region with a high probability of containing electrons.
What is the electron cloud rule?
The term electron cloud is used to describe the area around an atomic nucleus where an electron will probably be. Since we are discussing a very small (microscopic) system, an electron must be described using quantum mechanical rules rather than the classical rules which govern planetary motion. …
What is distort in chemistry?
ANY CHANGE IN LENGTHS OF CHEMICAL BONDS, AND IN THE ANGLES BETWEEN THEM, CAUSED BY STERIC HINDRANCE AND SIMILAR FORCES.
Why is a larger electron cloud more Polarizable?
Generally, polarizability increases as the volume occupied by electrons increases. In atoms, this occurs because larger atoms have more loosely held electrons in contrast to smaller atoms with tightly bound electrons. On rows of the periodic table, polarizability therefore decreases from left to right.
What does distort mean in chemistry?
Bond-distortion meaning (chemistry) Any change in the lengths of chemical bonds, and in the angles between them, caused by steric hindrance and similar forces.
What is wrong with the electron cloud model?
Explaining the behavior of these electron “orbits” was a key issue in the development of quantum mechanics. The electron cloud model says that we cannot know exactly where an electron is at any given time, but the electrons are more likely to be in specific areas.
Is the electron cloud dense?
Where the cloud is most dense, the probability of finding the electron is greatest; and where the electron is less likely to be, the cloud is less dense. These dense regions are known as “electron orbitals”, since they are the most likely location where an orbiting electron will be found.
What happens to electron clouds when a bond is formed?
If the molecules approach each other with sufficient energy, however, they may exchange atoms or actually form a bond (chemical reaction). Otherwise, their electron clouds will just deform to avoid the large electrostatic repulsion, and the two will then fly apart again.”
Why does Jahn Teller distortion occur?
The underlying cause of the Jahn–Teller effect is the presence of molecular orbitals that are both degenerate and open shell (i.e., incompletely occupied). This situation is not unique to coordination complexes and can be encountered in other areas of chemistry.
What is distortion of the electron distribution of an atom or molecule brought about by an ion or a polar molecule?
In a larger atom or molecule, the valence electrons are, on average, farther from the nuclei than in a smaller atom or molecule. They are less tightly held and can more easily form temporary dipoles. The ease with which the electron distribution around an atom or molecule can be distorted is called the polarizability.



