Why does atomic size increase down a group?
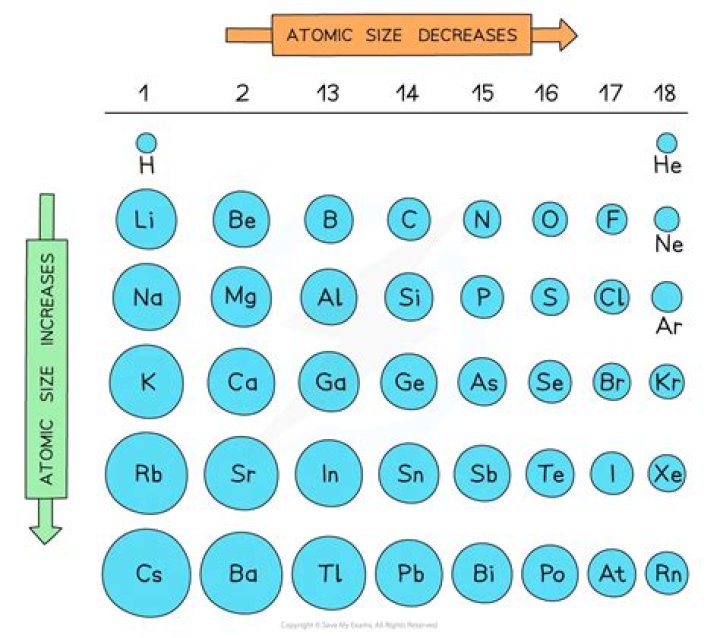
Down a group, the number of energy levels (n) increases, so there is a greater distance between the nucleus and the outermost orbital. This results in a larger atomic radius.
Why does the atomic size increase down a group class 10?
The growth of nuclear charge pulls more intensely the electrons, pulling them closer to the nucleus. The number of energy levels (n) increases in a group downwards, since there is a larger distance between the nucleus and the outermost orbital. This results in an atomic radius that is greater.
Why does atomic size increase as you go down a group quizlet?
Why does atomic radii increase as you move down a group? The number of energy levels increases as you move down a group as the number of electrons increases. Each subsequent energy level is farther from the nucleus than the previous one. Therefore, the atomic radius increases as the group and energy levels increase.
How does atomic size increase?
Summary: Atomic size increases as you go down a column because of the addition of another electron shell and electron shielding. Atomic size decreases as you go right across a row because of increased protons.
How does the atomic size vary down the group and why?
Atomic size of an element increases down a group. This is because as one moves down a group its number of electrons increases(not arithmetically), so number of shells also increases. As a result of which atomic size increases.
Why does atomic size generally increase down a group on the periodic table multiple choice question?
Why does atomic size generally increase as you move down a group of the periodic table and decrease as you move from left to right across a period? As you go from top to bottom, your increasing 1 energy level and the shielding affect comes into place, allowing atomic size to increase.
Why does the atomic size increase as you move top to bottom in a group?
This is caused by the increase in the number of protons and electrons across a period. One proton has a greater effect than one electron; thus, electrons are pulled towards the nucleus, resulting in a smaller radius. Atomic radius increases from top to bottom within a group.
Why does atomic size decrease when moving to the right across a period?
Atomic size gradually decreases from left to right across a period of elements. This means that the nucleus attracts the electrons more strongly, pulling the atom’s shell closer to the nucleus. The valence electrons are held closer towards the nucleus of the atom. As a result, the atomic radius decreases.
How does atomic size increase or decrease?
Moving Across the Periodic Table. an increase in atomic size because of additional repulsions between electrons, a decrease in size because of the additional protons in the nucleus, no effect at all as the two opposing tendencies of electron repulsion and nuclear attraction balance each other out.
Why does atomic size decreases along the period?
Within a period, protons are added to the nucleus as electrons are being added to the same principal energy level. These electrons are gradually pulled closer to the nucleus because of its increased positive charge. Since the force of attraction between nuclei and electrons increases, the size of the atoms decreases.
Why does the size of atoms increase going down a group?
Explain why the size of atoms increases going down a group on the periodic table. The atomic radii of elements in the same period have the trend of decreasing while moving from left to right of the periodic table. Also, the atomic radii of elements in the same group follows the trend of increasing while moving from top to bottom of the group.
What is the relationship between atomic radius and nuclear charge?
In general, the atomic radius decreases as we move from left to right in a period with an increase in nuclear charge of the element. The atomic radius increases when we go down a group because of the addition of extra shell.
Why does the radius of an atom change across a period?
This results in a DECREASE in atomic radii across the Period, due to the increased nuclear charge which draws in the valence electrons. On the other hand, going down a Group, we go to another so-called shell of electrons, that build on the preceding shell.
How do you define atomic size?
Define atomic size. How does it vary in a period and in a group? Atomic size is the distance between the centre of the nucleus of an atom and its outermost shell and the atomic radius is defined as theshortest distance between the atom’s nuclei and the outermost shell of the atom.



