Why is benzyl group preffered in hydrogenolysis?
The main interest in hydrogenolysis of benzyl derivatives stems from the use of the benzyl group as a protecting group for hydroxy, amino and carboxylic acids. In many hydrogenolysis reactions the presence of an acid (<1%) enhances the reaction rate.
What is the reduction product of hydrogenolysis?
Hydrogenolysis is a chemical reaction whereby a carbon–carbon or carbon–heteroatom single bond is cleaved or undergoes lysis (breakdown) by hydrogen. The heteroatom may vary, but it usually is oxygen, nitrogen, or sulfur.
Is hydrogenolysis a reduction?
Hydrogenolysis refers to the cleavage of carbon-carbon and various other carbon bonds in the reduction conditions and the substitution of the released atoms or groups with hydrogen atoms [117].
Which catalyst is used in hydrogenolysis?
Hydrogenation is a chemical reaction between molecular hydrogen (H2) and another compound or element, usually in the presence of a catalyst such as nickel, palladium or platinum. The process is commonly employed to reduce or saturate organic compounds.
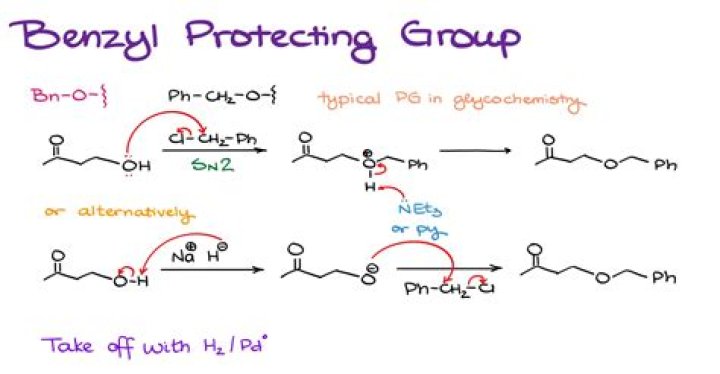
What is the benzyl group in chemistry?
In organic chemistry, benzyl is the substituent or molecular fragment possessing the structure C6H5CH2–. Benzyl features a benzene ring attached to a CH2 group.
What is hydrogenolysis example?
In petroleum refineries, catalytic hydrogenolysis of feedstocks is conducted on a large scale to remove sulfur from feedstocks, releasing gaseous hydrogen sulfide (H2S). The hydrogen sulfide is subsequently recovered in an amine treater and finally converted to elemental sulfur in a Claus process unit.
What do you mean by hydrogenolysis?
Hydrogenolysis is a catalytic chemical reaction that breaks a chemical bond in an organic molecule with the simultaneous addition of a hydrogen atom to the resulting molecular fragments.
What is the formula of benzoyl?
C₁₄H₁₀O₄
Benzoyl peroxide/Formula
Which of the following molecule is benzyl?
The molecule consists of a benzene ring substituted with a bromomethyl group. It is a colorless liquid with lachrymatory properties. The compound is a reagent for introducing benzyl groups….Benzyl bromide.
| Names | |
|---|---|
| ECHA InfoCard | 100.002.589 |
| IUPHAR/BPS | 6294 |
| PubChem CID | 7498 |
| UNII | XR75BS721D |
How do you Deprotect benzyl group?
Benzyl protecting groups can be removed using a wide range of oxidizing agents including:
- CrO3/acetic acid at ambient temperature.
- Ozone.
- N-Bromosuccinimide (NBS)
- N-Iodosuccinimide (NIS)
What is the example of hydrogenolysis?
What is the difference between hydrogenation and hydrogenolysis?
Hydrogenation refers to the reaction between a susbtance and molecular hydrogen H2 . Hydrogenolysis refers to the breaking of a bond between two carbon atoms or between an atom of carbon and that of another element via reaction with hydrogen.



