Why is quenching important chemistry?
Quenching refers to any process which decreases the fluorescence intensity of a given substance. A variety of processes can result in quenching, such as excited state reactions, energy transfer, complex-formation and collisional quenching. Molecular oxygen, iodide ions and acrylamide are common chemical quenchers.
How does quenching affect fluorimetry?
Abstract. Fluorescence quenching refers to any process that decreases the fluorescence intensity of a sample. A variety of molecular interactions can result in quenching. These include excited-state reactions, molecular rearrangements, energy transfer, ground-state complex formation, and colli-sional quenching.
Why is fluorescence quenching important?
Fluorescence quenching is an important technique for measuring binding affinity between ligands and proteins. Fluorescence quenching is the decrease in the quantum yield of fluorescence from a fluorophore, induced by a variety of molecular interactions with quencher molecule(s).
What does quenching mean in organic chemistry?
quench. (…) (transitive, chemistry) To terminate or greatly diminish (a chemical reaction) by destroying or deforming the remaining reagents.
What are the disadvantages of quenching steel?
Pros and Cons of Metal Quenching However, water does have a few drawbacks when it comes to quenching. This method can lead to distortion of the metal, as well as cracking. If this happens, the material becomes compromised and may end up useless. Oils such as mineral or cottonseed are also frequently used for quenching.
Why is oxygen a good quencher?
Oxygen is one of the most powerful luminescence quenchers. Moreover, the energies of excited states of oxygen (1∑g+ and 1Δg) are lower than the energies of the excited states of most organic dyes and metal complexes (Figure 1.1), which makes quenching via energy transfer favourable.
What is a quenching problem?
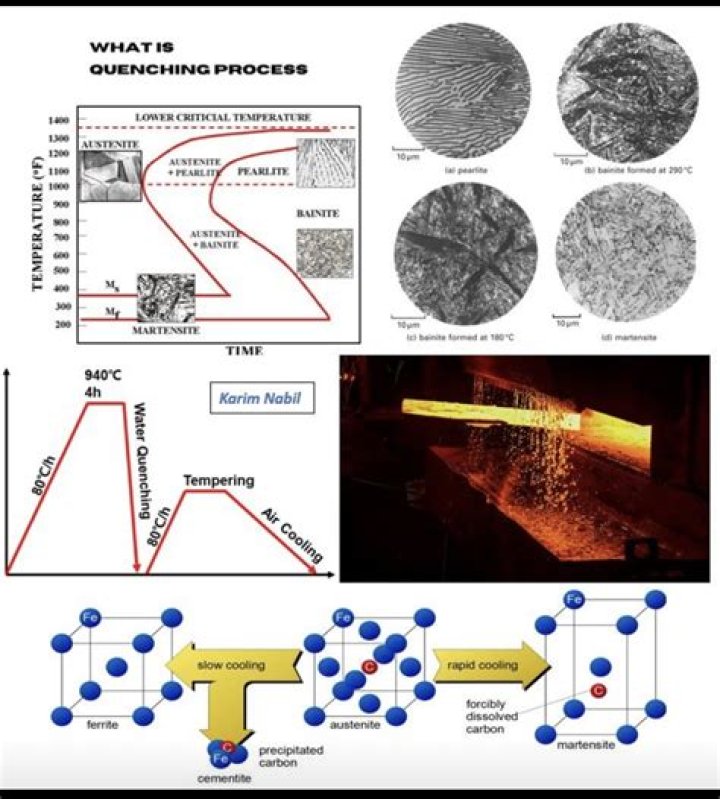
Often, after quenching, an iron or steel alloy will be excessively hard and brittle due to an overabundance of martensite. In these cases, another heat treatment technique known as tempering is performed on the quenched material in order to increase the toughness of iron-based alloys.
What is aggregation caused quenching?
Aggregation-caused quenching (ACQ) is a known phenomenon responsible for low emission of dyes when in the form of solid state (e.g. in nanostructures, nano-precipitates), although in solution (in appropriate solvent) these are characterized with bright emission [27,28].
What happens in fluorescence quenching?
Fluorescence quenching is a physicochemical process that lowers the intensity of emitted light from fluorescent molecules. When a molecule absorbs light, electrons in its constituent atoms become excited and are promoted to a higher energy level.
How does quenching affect microstructure?
The rapid quenching changes the crystal structure of the steel, compared with a slow cooling. Depending on the carbon content and alloying elements of the steel, it can get left with a harder, more brittle microstructure, such as martensite or bainite, when it undergoes the quench hardening process.
Does quenching make metal brittle?
Quenching results in a metal that is very hard but also brittle. Gently heating a hardened metal and allowing it to cool slowly will produce a metal that is still hard but also less brittle.
What is an example of quenching?
Quenching (fluorescence) A variety of processes can result in quenching, such as excited state reactions, energy transfer, complex-formation and collisional quenching. As a consequence, quenching is often heavily dependent on pressure and temperature. Molecular oxygen, iodide ions and acrylamide are common chemical quenchers.
What is fluorescence quenching in chemistry?
Fluorescence Quenching. Summary. The emission of light from the excited state of a molecule ( uorescence or phospho- rescence) can be quenched by interaction with another molecule.
What is analytical chemistry?
Analytical Chemistry is a journal covering the categories related to Analytical Chemistry (Q1). It is published by American Chemical Society. The overall rank of Analytical Chemistry is 1200. ISSN of this journal is/are 15206882, 00032700 . Impact Score: 6.66 h-Index: 332 SJR: 2.117 Overall Ranking: 1200
What is the ISSN of annual review of analytical chemistry?
Annual Review of Analytical Chemistry is a journal covering the categories related to Analytical Chemistry (Q1); Medicine (miscellaneous) (Q1). It is published by Annual Reviews Inc.. The overall rank of Annual Review of Analytical Chemistry is 641. ISSN of this journal is/are 19361335, 19361327 .



