Why is tropylium ion stable?
It attains the extra stability due to the conjugation of positive charges with the pi bonds. It has seven resonating structures. The more number of the resonating structure increases its stability concerning benzylic cation. Along with the resonance, the Tropylium ions follow the Huckle rule of aromaticity.
Why is Tropylium aromatic?
– The molecule must be cyclic that is, a ring of atoms. Number of π electrons in the ring are 6, therefore, tropylium cation also follows Huckel’s Rule of (4n+2)π electrons, where n = 1. Thus, tropylium cation is satisfying all the conditions for aromaticity and consequently, aromatic in nature.
How tropylium ion is generated?
The tropylium ion arises from the parent ion of 1 (2) by either a one-step process, mle 148 (152) + mle 91 (93) for which a metastable ion (m*) appears at 55.9 (56.9) or by a two-step process in which CH,O is lost from the parent ion, giving mle 118 (120) followed by the elimination of a BO radial.
What is the structure of tropylium cation?
In organic chemistry, the tropylium ion or cycloheptatrienyl cation is an aromatic species with a formula of [C7H7]+….Tropylium cation.
| Names | |
|---|---|
| Chemical formula | C 7H + 7 |
| Molar mass | 91.132 g·mol−1 |
| Structure | |
| Point group | D7h |
Why tropylium anion is non aromatic?
tropylium ion is anti-aromatic. it has conjugation of 3 pi bonds(6e) and 2e from the negative charge. according to huckel’s rule due to 4n electrons conjugation it is antiaromatic.
Why tropylium bromide is an ionic compound not covalent?
7-membered rings- Cycloheptatriene It turns out that cycloheptatrienyl bromide is an ionic compound, since its cation (known as tropylium cation) is aromatic. In the covalent form, there is no continuity in p orbital overlap as one of the carbon atoms is sp3 hybridized.
Why Tropylium bromide is an ionic compound explain?
Answer: Trypolium bromide is an ionic compound due to which it is able to dissolve in water. Explanation: Trypolium bromide is present in nature as a salt which contains a trypolium cation and a bromine anion. Since, it contains ions so it is an polar compound.
Is Tropylium ion conjugated?
The tropylium anion really has two valence electrons on the negative carbon: Electron conjugation all around the ring.
Why Tropylium bromide is an ionic compound not covalent?
How many conjugated p orbitals does the Tropylium ion have?
The vacant p orbital does not change the pi electron count so the tropylium cation has 6 pi electrons which obeys the 4n + 2 rule for aromaticity. The molecular orbital diagram for the 7 p orbitals in the tropylium cation has three bonding MO’s and 4 antibonding MO’s.
What is Tropylium anion?
tropylium ion is anti-aromatic. it has conjugation of 3 pi bonds(6e) and 2e from the negative charge. according to huckel’s rule due to 4n electrons conjugation it is antiaromatic. it could be non aromatic as the anion is big and could be non planar, though it is planar and hence antiaromatic.
Why Tropylium anion is non aromatic?
What is tropylium ion in mass spectrometry?
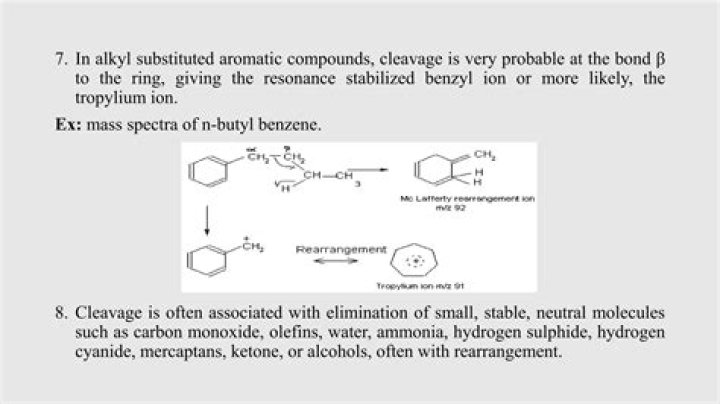
The tropylium ion is frequently encountered in mass spectrometry in the form of a signal at m/z = 91 and is used in mass spectrum analysis. This fragment is often found for aromatic compounds containing a benzyl unit. Upon ionization, the benzyl fragment forms a cation (PhCH +.
What is the equilibrium constant of tropylium?
The equilibrium constant is 1.8 × 10−5, making it about as acidic in water as acetic acid. The tropylium ion is frequently encountered in mass spectrometry in the form of a signal at m / z = 91 and is used in mass spectrum analysis. This fragment is often found for aromatic compounds containing a benzyl unit.
What is the ionic structure of tropylium bromide?
The structure of tropylium bromide was deduced to be a salt, C7H7+Br–, by Doering and Knox in 1954 by analysis of its infrared and ultraviolet spectra. The ionic structures of tropylium perchlorate (C7H7+ClO4–) and tropylium iodide (C7H7+I–) in the solid state have been confirmed by X-ray crystallography.
Is tropylium ion an acid or base?
The tropylium ion is an acid in aqueous solution (i.e., an Arrhenius acid) as a consequence of its Lewis acidity: it first acts as a Lewis acid to form an adduct with water, which can then donate a proton to another molecule of water: ( Boric acid gives acidic aqueous solutions in much the same way.)



