Why sigma bonds are stronger than pi bond?
In sigma bonds, the large overlap of the orbital involves the removal of a large amount of energy. While in pi bonds the extent of overlapping is less than sigma bond. Therefore, sigma bond is stronger than pi bond.
Why are sigma bonds the strongest?
Sigma bonds result from the head-on overlap of the orbitals of the two atoms, which has more area of overlap than pi bonds which result from the parallel overlap of orbitals. That is why sigma bonds are stronger.
How do sigma bond strengths compare?
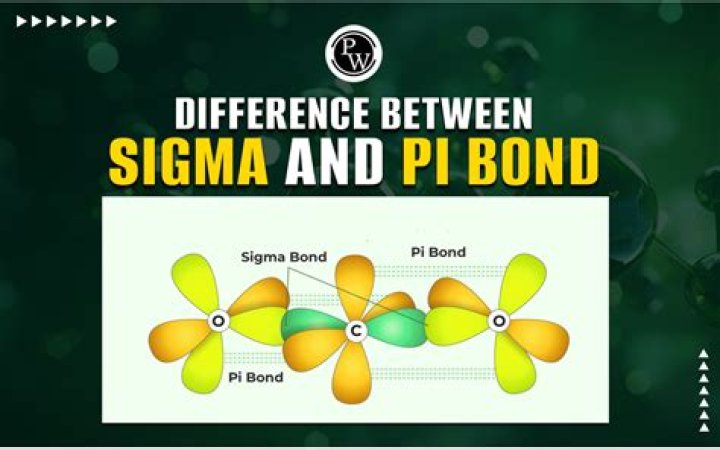
Difference Between Sigma Bond and Pi Bond
| Parameter | Sigma Bond |
|---|---|
| Bond Strength | The bond strength of sigma bonds is more than pi bonds. |
| Bond Forming Order | Sigma bonds are formed first when atoms come closer |
| Number of Bonds | There is only one sigma bond between two atoms. |
Is a sigma bond strong?
In chemistry, sigma bonds (σ bonds) are the strongest type of covalent chemical bond. They are formed by head-on overlapping between atomic orbitals.
Which sigma bond is the strongest?
Yes, generally p-p sigma bond is stronger than s-s sigma bond. This is because, bond strength is directly proportional to the directional nature of orbitals in overlapping.
Why is a sigma bond stronger than a pi bond quizlet?
Sigma bonds are significantly stronger than pi bonds. This is because sigma bonds allow for electron density to be concentrated to a much larger degree between the two nuclei. a single bond, or the first bond or a double or triple bond) and involves head to head overlap of two atomic orbitals.
Which is the strongest bonding?
covalent bond
In chemistry, covalent bond is the strongest bond. In such bonding, each of two atoms shares electrons that binds them together. For example, water molecules are bonded together where both hydrogen atoms and oxygen atoms share electrons to form a covalent bond.
How do you compare pi bond strengths?
Strength of Sigma and Pi Bond Strength of a bond basically depends on the extent of overlapping of atomic orbitals. Sigma bond form overlapping along the internuclear axis which is powerful than the pi bond which overlaps sideways. The area of overlap in pi bonds is lesser as compared to sigma bonds.
What is difference between Pi and sigma bond?
A sigma bond is formed by head-on overlapping of hybrid orbitals (along the bonding axis). A pi bond is formed by side-to-side overlapping of hybrid orbitals (above and below the bonding axis). It is denoted by the symbol σ. It is usually denoted by the symbol π.
Why is sigma bond stronger than pi Quora?
Since in formation of a sigma bond, the orbitals are along inter nuclear axis this results in more effective orbital overlap as compared to when pi bonds are formed as they are perpendicular to the internuclear axis. This significant difference in orbital overlap results in sigma bond being stronger than the pi bond.
Is Delta bond stronger than pi bond?
Delta bond is formed by d orbitals because in delta bond four lobe interaction is there . it is stronger than pie bond because 4 lobes are interacting here.
Which type of bond is weakest?
ionic bond
The ionic bond is generally the weakest of the true chemical bonds that bind atoms to atoms.
Why is a pi bond weaker than a sigma bond?
Pi bonds are usually weaker than sigma bonds. This is contrasted by sigma bonds which form bonding orbitals directly between the nuclei of the bondingatoms, resulting in greater overlap and a strong sigma bond. Pi bonds result from overlap of atomic orbitals that are in contact through two areas of overlap.
What are the differences between a sigma bond and a pi bond?
• Sigma bonds are formed by head to head overlapping of orbitals, whereas pi bonds are formed by the lateral overlapping. • Sigma bonds are stronger than pi bonds. • Sigma bonds can be formed between both s and p orbitals whereas pi bonds are mostly formed between p and d orbitals.
Are sigma bonds more electrophilic than pi bonds?
Sigma bonds have more s-character, are closer to the nucleus, and are stronger than pi-bonds. These two factors combine to make the carbonyl functional group a good electrophilic species. The movie below shows a nice summary of a general nucleophilic attack onto a C=O bond:
How do you determine Sigma and pi bonds?
Usually, all bonds between atoms in most organic compounds contain one sigma bond each. If it is a single bond, it contains only sigma bond. Double bonds have one each, and triple bonds have one sigma bond and two pi bonds.



